A comparative dosimetric study of seven radiation techniques for breast cancer after mastectomy and immediate breast reconstruction
Introduction
Postmastectomy radiotherapy (PMRT) can reduce the locoregional recurrence rate and improve overall survival in high risk breast cancer patients (1,2). Breast reconstruction can improve the cosmetic effect and quality of life of patients after mastectomy, and more than 50% of breast cancer patients receive breast reconstruction after mastectomy (3,4). However, the optimal mode of breast reconstruction and PMRT is still controversial.
Breast reconstruction after mastectomy includes autologous breast reconstruction and breast reconstruction with implant. PMRT did not have adverse effects on reconstruction in several studies with autologous breast reconstruction (5,6). However, studies on breast reconstruction with an implant suggested that severe capsular contracture occurred after PMRT, which may cause implant loss (7-11). A previous study has been reported that the exposure dose to breast implant is one important factor for capsular contracture (12), but relevant research to identify the optimal irradiation technique to reduce the exposure dose to breast implant is lacking.
At present, conventional PMRT is still a tangential field technique based on three-dimensional conformal radiation therapy (3DCRT). Modern radiotherapy techniques with better dose homogeneity and conformity, including intensity-modulated radiation therapy (IMRT) and volumetric-modulated arc therapy (VMAT) have been adopted in clinical research of breast cancer (13-17). Additionally, the application of flattening filter-free (FFF) beams in IMRT can achieve a high-dose rate, and reduce the treatment time and dose to surrounding normal tissues (18). In this study, we carried out the dosimetric comparison to breast implant after mastectomy among seven different radiation techniques, including 3DCRT, field-in-field IMRT (FIF-IMRT), the mixture of 3DCRT and IMRT (HYBRID), IMRT, FFF-IMRT, VMAT and FFF-VMAT, and explored the optimal radiation technique for breast cancer patients after mastectomy and immediate breast reconstruction.
Methods
Patients
Ten breast cancer patients between July 2011 and December 2013 were identified from the First Affiliated Hospital of Xiamen University in the current study. Patients were included if they met the following criteria: (I) female, unilateral breast cancer, received mastectomy and axillary lymph node dissection, and immediate permanent implant breast reconstruction; (II) stage of II or III (T1-4N1-3M0) according to the 7th edition (2009) of the American Joint Committee on Cancer/Union for International Cancer Control staging system. The study was approved by the ethics committee of the First Affiliated Hospital of Xiamen University (approval number of institutional review board, 2016J01635). Patient characteristics are showed in Table 1.

Full table
Target volume and organ at risks (OARs) delineation
The delineation of the clinical target volume (CTV) and OARs was finally approved by three experienced radiation oncologists. Since this study was a dosimetric comparison, CTV was delineated as the ipsilateral chest wall. The ipsilateral chest wall was delineated according to the recommendations of the International Commission on Radiation Units report 83 (19). The cranial, caudal, medial and lateral borders for CTV were the bottom of the clavicle head, a 1 cm margin from below the contralateral breast, the ipsilateral sternal-rib junction, and the mid-axillary line, respectively. The planning target volume (PTV) was defined as a 5-mm expansion in all directions around the CTV except for the skin surface, including the set-up margin and accounting for patient movement.
Delineation of OARs included the breast implant, contralateral breast, ipsilateral lung, contralateral lung, and heart on the computed tomography image of each slice. Table 2 lists the volume of the target volume and normal tissue of the 10 patients.

Full table
Radiation techniques
The prescription dose to the PTV was 50 Gy with 2.0 Gy per fraction in 25 fractions. The radiation plans used six MV photon beams. Seven radiation techniques were generated and calculated to compare the difference of dosimetry in the PTV, breast implant and OARs, including the ipsilateral lung and heart in left breast cancer. Treatment planning was performed in Eclipse (Varian Medical Systems, PRO 11.0, AAA 11.0) treatment planning system. All plans were normalized in such that at least 95% of the PTV received 95% of the prescribed dose.
3DCRT
A paired, conventional opposed tangential technique with a physical wedge was applied.
FIF-IMRT
Briefly, the contribution of two opposed tangential beams and multiple subfields were used to achieve the dose homogeneity of the PTV without wedges. The dose distributions of an open beam configuration were first calculated and evaluated. The lung block was formed by multileaf collimator (MLC) and then used to smooth out the lateral hot spots. Additional 2–3 subfields were generated using by manually fitting the MLC to ‘‘hot’’ areas.
IMRT
Five tangential direction fields were generated for homogeneous dose delivery to the PTV using a dynamic sliding window MLC. An angle of 20° to 30° separated the two beams, which were oriented in the same direction. The maximum number of segments was 50.
HYBRID
In the HYBRID plan, a mixture of 3DCRT and IMRT techniques was used. 3DCRT technique (40 Gy/20f) was used for the first 20 times, and IMRT (10 Gy/5f) for the later five times.
FFF-IMRT
The tangential directions of FFF-IMRT were similar to IMRT using 6MV-X FFF beams.
VMAT
Two arcs were used for the VMAT plan. The first arc started from 300° to 170°. The second arc had exactly opposite starting and ending angles relative to the first arc.
FFF-VMAT
The two arcs of FFF-VMAT were similar to the VMAT plans by using 6MV-X FFF beams.
Conformity index (CI) and homogeneity index (HI) calculation
The CI and HI were evaluated according to our previous study (20). CI = (VTref/VT) × (VTref/Vref), where VTref represents the target volume covered by isodose, VT is the target volume and Vref is the total volume covered by 95% of isodose. CI range was 0–1, in which the conformity was better when the CI value was larger. HI = D5/D95, where D5 represents the radiation dose received by 5% of PTV, while D95 represents the radiation dose received by 95% of PTV. The closer the HI value is to 1, the better the target uniformity will be.
Statistical analysis
One-way analysis of variance tests was performed to compare dosimetric differences between the rival techniques. All statistical tests were performed using SPSS software (release 17.0, SPSS Inc., Chicago, IL, USA) and the differences were considered statistically significant at a P value of <0.05.
Results
Target volume coverage
Table 3 shows the target volume coverage of 10 patients. On the basis of meeting 95% of the coverage, the V105% of IMRT was minimal, significantly lower than that of 3DCRT and FIF-IMRT, but was not significantly different with FFF-IMRT. The Dmax of 3DCRT was maximal, significantly higher than that of HYBRID, which was the minimum, and significantly lower than that of VMAT and FFF-VMAT, but without differences with IMRT.

Full table
The CIs of 3DCRT, FIF-IMRT and HYBRID were significantly worse than those of IMRT, FFF-IMRT, VMAT, and FFF-VMAT. The CIs in IMRT, FFF-IMRT, VMAT, and FFF-VMAT were no significant difference. The HIs of IMRT and FFF-IMRT were superior to those of other techniques.
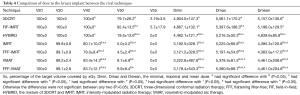
Doses to breast implant
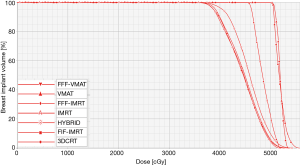
There were no significant differences in V20 (Vx indicating the volume of OARs or target volume receiving xGy), V30 and V55 among the seven techniques; the V40 and V50 values of IMRT, FFF-IMRT, VMAT, and FFF-VMAT were significantly lower than those of other techniques, but there was no significant difference between the four techniques. The IMRT and FFF-IMRT techniques could reduce the exposure dose and volume to breast implant including V40, V50, Dmax, and Dmean; significantly lower than those of 3DCRT and FIF-IMRT. The V40, V50, Dmin, and Dmean in IMRT and FFF-IMRT were also significant difference with HYBRID. IMRT and FFF-IMRT also had significantly lower dose to breast implant compared to VMAT and FFF-VMAT, but there was no significant difference between IMRT and FFF-IMRT (Table 4) (Figures 1,2).
Full table
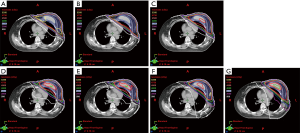
Dose to ipsilateral lung and heart
The V10 and Dmean of ipsilateral lung in VMAT and FFF-VMAT were higher than those of other techniques. In addition, there was no significant difference in the ipsilateral lung between VMAT and FFF-VMAT. The V10, V20, and Dmean of ipsilateral lung were also no significant difference among the techniques including 3DCRT, FIF-IMRT, HYBRID, and IMRT. FIF-IMRT was better than FFF-IMRT, VMAT, FFF-VMAT in terms of the V10. The dose to ipsilateral lung in IMRT was in the range of traditional radiation techniques and VMAT, but there was no significant difference between IMRT and FFF-IMRT (Table 5). The FFF technique did not affect the dose to the ipsilateral lung in IMRT and VMAT

Full table
For the exposure dose to the heart in seven patients with left breast cancer, the V10 of IMRT, FFF-IMRT, VMAT, and FFF-VMAT were significantly higher than that of other techniques. However, there were no significant differences among the four techniques including V10, V20, V30, V40, Dmin, Dmax and Dmean. In addition, the V40 of the heart were improved in IMRT, VMAT and FFF-VMAT compared with 3DCRT, FIF-IMRT, and HYBRID. The FFF technique also did not affect the dose to the heart in IMRT and VMAT (Table 5).
Discussion
3DCRT is generally recommended for radiation in breast cancer, but study has found that use of IMRT significantly reduce the radiation dose to OARs, with a better target dose coverage when compared to 3DCRT (17). In this study, we compared the target volume coverage, doses to breast implant and OARs using seven techniques for breast cancer after mastectomy and immediate breast reconstruction. Our results show that the IMRT technique minimizes the dose to breast implant, with acceptable doses to ipsilateral lung and heart, while ensuring a better target volume coverage.
The potential effect of PMRT on breast implant could lead to capsular contracture, which might even require removal of the breast implant (7-11). The exact correlation between PMRT and capsular contracture is still unclear. However, a previous study has found that PMRT can induce modifications of silicone (12), and radiotherapy is an important factor leading to breast implant removal (21,22). Nevertheless, unified criteria on the radiotherapy mode and technique after implant-based breast reconstruction are lacking. The use of PMRT for patients with positive axillary lymph nodes has increased over time, but the breast reconstruction rates did not decrease (23). Breast reconstructions are located in the conventional tangential field during PMRT. Therefore, it is difficult to avoid an irradiation dose to the breast reconstruction (24).
In a study with flap reconstruction, Lee et al. found that HYBRID could reduce over 10% of the mean dose to the flapped area compared with 3DCRT and FIF-IMRT, and the mean dose of HYBRID was 12.8% and 3.9% higher than that of IMRT and VMAT (25). Our results suggest that IMRT and FFF-IMRT techniques could reduce the exposure dose and volume to breast implant than those of other techniques. However, it is still unclear whether dose improvement can enhance the reconstruction effect. Ribuffo et al. found that radiation-induced modifications of silicone may be one of the co-factors underlying capsular contracture (12). Therefore, the exposure dose to breast implant should be reduced as much as possible, which may decrease the risk of capsular contracture. IMRT technique is suitable for use in breast cancer after mastectomy and immediate breast reconstruction.
FFF beams offer the potential benefit for a higher dose rate, shorter treatment time, and lower peripheral dose (18). In this study, we did not find that the application of FFF technique in IMRT and VMAT could improve the dose to the target volume, breast implant and OARs. Other studies have also failed to find the advantages of FFF technique in breast cancer radiation therapy (18,26), although FFF mode may be suitable for use with hypofractionated dose schemesJeny (27). IMRT exhibited acceptable acute toxicities and better clinical outcomes in patients after breast-conserving surgery (13,14,16). In our study, VMAT had advantages such as a better CI and significantly reduced high-dose range of OARs; however, the low-dose volume of VMAT was significantly higher than that of other techniques, especially in the ipsilateral lungs. In a dosimetric study of left sided breast cancer after breast-conserving surgery, the IMRT technique has demonstrated the combined advantages in target volume coverage and dose to most OARs, besides for the heart and coronary artery compared to VMAT (28). Therefore, the potential risk of radiation-induced secondary lung cancer should be considered before using VMAT (29), especially in young women with breast cancer.
Several limitations should be acknowledged in this study. First, it was a dosimetric study and unable to represent final clinical results. Second, movement caused by breathing could have affected the actual dose to the target volume and OARs during breast radiation, whereas an active breathing control (ABC) technique may have improved the relevant conditions (30,31). However, studies with breast IMRT after breast-conserving surgery in which the ABC technique was not applied have exhibited acceptable acute toxicities and clinical outcomes (13,14).
Conclusions
In conclusion, according to our dosimetric study, IMRT achieves similar or superior target volume coverage and a better breast implant sparing for patients with breast implant after immediate breast reconstruction.
Acknowledgments
The authenticity of this article has been validated by uploading the key raw data onto the Research Data Deposit (RDD) public platform (
Funding: This work was supported by the Sci-Tech Office of Guangdong Province (No. 2013B021800157, 2013B021800458) and the Natural Science Foundation of Fujian Province (No. 2016J01635).
Footnote
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/tcr.2017.06.38). The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. The study was conducted in accordance with the Declaration of Helsinki (as revised in 2013). The study was approved by the ethics committee of the First Affiliated Hospital of Xiamen University (No.2016J01635) and written informed consent was obtained from all patients.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- EBCTCG (Early Breast Cancer Trialists' Collaborative Group). Effect of radiotherapy after mastectomy and axillary surgery on 10-year recurrence and 20-year breast cancer mortality: meta-analysis of individual patient data for 8135 women in 22 randomised trials. Lancet 2014;383:2127-35. [Crossref] [PubMed]
- Wu SG, He ZY, Li Q, et al. Predictive value of breast cancer molecular subtypes in Chinese patients with four or more positive nodes after postmastectomy radiotherapy. Breast 2012;21:657-61. [Crossref] [PubMed]
- Parker PA, Youssef A, Walker S, et al. Short-term and long-term psychosocial adjustment and quality of life in women undergoing different surgical procedures for breast cancer. Ann Surg Oncol 2007;14:3078-89. [Crossref] [PubMed]
- Rowland JH, Desmond KA, Meyerowitz BE, et al. Role of breast reconstructive surgery in physical and emotional outcomes among breast cancer survivors. J Natl Cancer Inst 2000;92:1422-29. [Crossref] [PubMed]
- Kelley BP, Ahmed R, Kidwell KM, et al. A systematic review of morbidity associated with autologous breast reconstruction before and after exposure to radiotherapy: are current practices ideal? Ann Surg Oncol 2014;21:1732-8. [Crossref] [PubMed]
- Chang EI, Liu TS, Festekjian JH, et al. Effects of radiation therapy for breast cancer based on type of free flap reconstruction. Plast Reconstr Surg 2013;131:1e-8e. [Crossref] [PubMed]
- Cordeiro PG, Albornoz CR, McCormick B, et al. The impact of postmastectomy radiotherapy on two-stage implant breast reconstruction: an analysis of long-term surgical outcomes, aesthetic results, and satisfaction over 13 years. Plast Reconstr Surg 2014;134:588-95. [Crossref] [PubMed]
- Ho AL, Bovill ES, Macadam SA, et al. Postmastectomy radiation therapy after immediate two-stage tissue expander/implant breast reconstruction: a University of British Columbia perspective. Plast Reconstr Surg 2014;134:1e-10e. [Crossref] [PubMed]
- Shah C, Kundu N, Arthur D, et al. Radiation therapy following postmastectomy reconstruction: a systematic review. Ann Surg Oncol 2013;20:1313-22. [Crossref] [PubMed]
- Cordeiro PG, Pusic AL, Disa JJ, et al. Irradiation after immediate tissue expander/implant breast reconstruction: outcomes, complications, aestheticresults, and satisfaction among 156 patients. Plast Reconstr Surg 2004;113:877-81. [Crossref] [PubMed]
- Nava MB, Pennati AE, Lozza L, et al. Outcome of different timings of radiotherapy in implant-based breast reconstructions. Plast Reconstr Surg 2011;128:353-9. [Crossref] [PubMed]
- Ribuffo D, Lo Torto F, Giannitelli SM, et al. The effect of post-mastectomy radiation therapy on breast implants: Unveiling biomaterial alterations with potential implications on capsular contracture. Mater Sci Eng C Mater Biol Appl 2015;57:338-43. [Crossref] [PubMed]
- Yang Z, Zhang L, Chen X, et al. Multibeam inverse intensity-modulated radiotherapy (IMRT) for whole breast irradiation: a single center experience in China. Oncotarget 2015;6:35063-72. [PubMed]
- Ha B, Suh HS, Lee J, et al. Long-term results of forward intensity-modulated radiation therapy for patients with early-stage breast cancer. Radiat Oncol J 2013;31:191-8. [Crossref] [PubMed]
- Scorsetti M, Alongi F, Fogliata A, et al. Phase I-II study of hypofractionated simultaneous integrated boost using volumetric modulated arc therapy for adjuvant radiation therapy in breast cancer patients: a report of feasibility and early toxicity results in the first 50 treatments. Radiat Oncol 2012;7:145. [Crossref] [PubMed]
- Fiorentino A, Mazzola R, Ricchetti F, et al. Intensity modulated radiation therapy with simultaneous integrated boost in early breast cancer irradiation. Report of feasibility and preliminary toxicity. Cancer Radiother 2015;19:289-94. [Crossref] [PubMed]
- Fiorentino A, Ruggieri R, Giaj-Levra N, et al. Three-dimensional conformal versus intensity modulated radiotherapy in breast cancertreatment: is necessary a medical reversal? Radiol Med 2017;122:146-53. [Crossref] [PubMed]
- Spruijt KH, Dahele M, Cuijpers JP, et al. Flattening filter free vs flattened beams for breast irradiation. Int J Radiat Oncol Biol Phys 2013;85:506-13. [Crossref] [PubMed]
- ICRU Report 83. Prescribing, Recording, and Reporting Photon-Beam Intensity-Modulated Radiation Therapy (IMRT). Bethesda: International Commission on Radiation Units and Measurements, 2010.
- He Z, Wu S, Zhou J, et al. Accelerated partial breast irradiation with intensity-modulated radiotherapy is feasible for chinese breast cancer patients. J Breast Cancer 2014;17:256-64. [Crossref] [PubMed]
- Anker CJ, Hymas RV, Ahluwalia R, et al. The Effect of Radiation on Complication Rates and Patient Satisfaction in Breast Reconstruction using Temporary Tissue Expanders and Permanent Implants. Breast J 2015;21:233-40. [Crossref] [PubMed]
- Jagsi R, Jiang J, Momoh AO, et al. Complications After Mastectomy and Immediate Breast Reconstruction for Breast Cancer: A Claims-Based Analysis. Ann Surg 2016;263:219-27. [Crossref] [PubMed]
- Frasier LL, Holden S, Holden T, et al. Temporal Trends in Postmastectomy Radiation Therapy and Breast Reconstruction Associated With Changes in National Comprehensive Cancer Network Guidelines. JAMA Oncol 2016;2:95-101. [Crossref] [PubMed]
- Motwani SB, Strom EA, Schechter NR, et al. The impact of immediate breast reconstruction on the technical delivery of postmastectomy radiotherapy. Int J Radiat Oncol Biol Phys 2006;66:76-82. [Crossref] [PubMed]
- Lee JA, Yoon WS, Chung SY, et al. Can intensity-modulated radiation therapy spare the central flapped area while encompassing the target volume in radiotherapy after immediate breast reconstruction? J Med Imaging Radiat Oncol 2013;57:595-602. [Crossref] [PubMed]
- Kretschmer M, Sabatino M, Blechschmidt A, et al. The impact of flattening-filter-free beam technology on 3D conformal RT. Radiat Oncol 2013;8:133. [Crossref] [PubMed]
- Bahrainy M, Kretschmer M, Jöst V, et al. Treatment of breast cancer with simultaneous integrated boost in hybrid plan technique: Influence of flattening filter-free beams. Strahlenther Onkol 2016;192:333-41. [Crossref] [PubMed]
- Zhao H, He M, Cheng G, et al. A comparative dosimetric study of left sided breast cancer after breast-conserving surgerytreated with VMAT and IMRT. Radiat Oncol 2015;10:231. [Crossref] [PubMed]
- Filippi AR, Ragona R, Piva C, et al. Optimized volumetric modulated arc therapy versus 3D-CRT for early stage mediastinal Hodgkin lymphoma without axillary involvement: a comparison of second cancers and heart disease risk. Int J Radiat Oncol Biol Phys 2015;92:161-8. [Crossref] [PubMed]
- Remouchamps VM, Vicini FA, Sharpe MB, et al. Significant reductions in heart and lung doses using deep inspiration breath hold with active breathing control and intensity-modulated radiation therapy for patients treated with locoregional breast irradiation. Int J Radiat Oncol Biol Phys 2003;55:392-406. [Crossref] [PubMed]
- Chi F, Wu S, Zhou J, et al. Dosimetric comparison of moderate deep inspiration breath-hold and free-breathing intensity-modulated radiotherapy for left-sided breast cancer. Cancer Radiother 2015;19:180-6. [Crossref] [PubMed]