
Esophageal metastasis from endometrial adenocarcinoma: a case report and literature review
Introduction
Involvement of the esophagus by metastatic disease from primary malignancies is rare, occurring in 3–6% of patients, based on the investigation of long-term follow-up and post-mortem series (1-3). If local invasion from adjacent organs is excluded, this phenomenon is observed in only 1% of patients with malignant disease (4). It has been reported that primary tumors of stomach, breast, larynx, tongue, bronchus, prostate, tibia, liver, and pleura have a tendency to metastasize to the esophagus (2,3,5-7). However, metastasis from endometrial carcinoma is a very rare occurrence. Herein, we report the case of an endometrial adenocarcinoma involving the esophagus that was refractory to second-line chemotherapy but was responsive to the single agent apatinib. We also review the literature.
Case presentation
Patient data
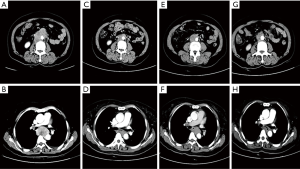
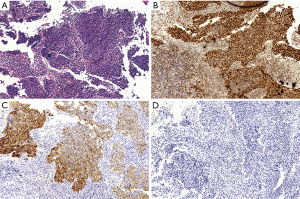
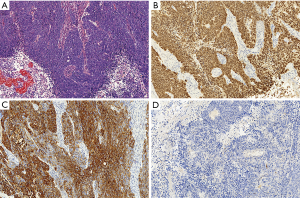
A 58-year-old woman underwent radical hysterectomy with bilateral salpingo-oophorectomy for endometrial adenocarcinoma in April 2014. Following this surgery, no radiation therapy and chemotherapy were administrated. She remained well until September 2016, when she was admitted to our hospital for progressive dysphagia and some lower abdominal pain for 3 months. A CT scan obtained at that time revealed a mass in the mid-esophagus (Figure 1) and esophagoscopy revealed a protruding lesion covered with normal esophageal mucosa 28 cm from the incisors. The abdominal CT scan demonstrated multiple enlarged lymph nodes in retroperitoneum and pelvic cavity (Figure 1). Subsequently, pathological and immunohistochemical analyses of the esophageal lesion biopsy indicated a low-differentiated adenocarcinoma with positive expression of cytokeratin (CK) 7, paired box gene 8 (PAX8), and negative expression of CK20. This result was consistent with the findings of endometrial adenocarcinoma, which had been removed 2 years earlier, demonstrating that the tumor was an esophageal metastasis from her previous endometrial adenocarcinoma (Figures 2,3).
Treatment procedures
After multidisciplinary consultation, the patient received 6 cycles of chemotherapy with paclitaxel (175 mg/m2, day 1, every 3 weeks) and carboplatin (50 mg/m2, day 1, every 3 weeks) as first-line chemotherapy from October 2016 to February 2017. During the treatment, the patient achieved partial remission (Figure 1) and symptoms of dysphagia were significantly relieved. However, in March 2017, progressive disease was detected as an augmented esophageal lesion on a CT scan (Figure 1). Despite administering epirubicin (85 mg/m2, day 1, every 3 weeks) combined with cisplatin (50 mg/m2, days 1–3, every 3 weeks) as second-line chemotherapy, a CT scan of the chest indicated disease progression after 2 cycles of chemotherapy. Hence, chemotherapy was suspended and the patient was started on apatinib (500 mg/day) in April, with shrinkage of the esophageal lesion and a stable lesion of the pelvic cavity within 2 months of starting therapy (Figure 1). To date, she has received targeted treatment over 6 months and has had stable disease, experiencing side effects of mild hand-foot syndrome and hypertension.
Discussion
Since the first reported case of metastatic esophagus carcinoma from the prostate by Gross and Freedman in 1942 (8), numerous other reports from a variety of tumors have been reported, including breast, larynx, thyroid, hypopharynx, and stomach, most of which have involved breast and lung cancer (6,9). Our case appears to be the second report of esophageal metastasis from an endometrial adenocarcinoma. The first case was reported by Zarian and associates in 1983 (10). It is difficult to obtain an accurate diagnosis of metastatic esophageal carcinomas from endometrial adenocarcinoma based only on clinical, endoscopic, radiological and histopathological features. Symptoms related to esophageal manifestations often lack specificity and generally occur several years or decades after the diagnosis and treatment of the primary lesion. Although endoscopic diagnosis is indispensable to the diagnostic examinations, the lesion generally presents as a normal-appearing mid-esophageal stricture, biopsies of which will be non-diagnostic (6,8-10). CT scan was only useful in the evaluation of the esophageal wall thickening and extra-esophageal mediastinal abnormalities (3).
Comprehensive immunohistochemical analyses may be the optimal method to differentiate between primary esophageal carcinoma and metastatic carcinoma from distant organs. The immunohistochemical analyses of endometrial adenocarcinoma are usually positive for CK7, PAX8 and negative for CK20 (11-13). Wang and associates demonstrated that the CK7−/CK20+ immunophenotype strongly suggests carcinomas from the gastrointestinal tract, particularly those of colorectal origin. The CK7+/CK20− immunophenotype indicated carcinomas from most other primary sites, including breast, non-mucinous ovarian, endometrial, lung carcinomas, and malignant mesothelioma (14). PAX8 is usually expressed in Müllerian tumors and is not expressed in many non-Müllerian tumors, including breast, colon, and gastroesophageal cancer (15). In our case, endoscopy revealed a mid-esophageal protruding lesion with pathology of a low-differentiated adenocarcinoma. Subsequently, immunohistochemical analyses of endometrial pathological sections were reappraised to obtain accurate diagnosis. The results were as follows: CK7(+), CK20(−) and PAX8(+). Hence, the pathologist easily diagnosed that the primary lesion as an endometrial adenocarcinoma because of the cell type and immunohistochemical analyses.
Secondary esophageal carcinoma occurs by three mechanisms: direct extension, involvement by mediastinal lymph node metastases, and hematogenous spread (2,3,9,10). A review of 62 cases of secondary esophageal involvement showed that direct extension from the contiguous or adjacent organs accounted for 45.2% of the cases, 35.5% were spread via mediastinal nodes, and 19.3% occurred by hematogenous spread (3). When the esophagus is involved by direct tumor extension, in most instances, the primary lesions are often located at the thyroid, pharynx, larynx, lung, and stomach (3,10). It is believed that lung, breast, and pancreas tumors metastasize to the esophagus via mediastinal lymph nodes (16). Hematogenous metastasis to the esophagus is infrequent and often occurs from carcinoma of the liver, kidney, uterus, skin, tongue, eye, bone, uterus and prostate (3,8,10). In our case, the endometrial adenocarcinoma is presumed to have spread by the hematogenous route because no extensive involvement of mediastinal lymph node was observed on CT scan.
As the clinical occurrence of esophageal carcinoma metastasis from distant primary organs occurs rarely, the diagnosis and treatment are often delayed and the treatment is not well standardized. As most patients with metastases to the esophagus already have metastases to other areas, the disease should be treated with a systemic approach (chemotherapy, biological therapy, radiotherapy or combination of these) (2). According to previous treatment modalities for metastatic carcinoma, especially those with a solitary metastatic lesion, surgical intervention should be performed for palliation or certain cases of solitary resectable metastatic lesions, which have demonstrated a good result, such as breast cancer, lung cancer, and malignant melanoma (2,6,17). Shimada and colleagues advocated the resection of solitary metastatic esophageal lesions from breast cancer, and the patient achieved local control with a disease-free interval of approximately 5 years (17). Oka et al. also reported a case without recurrence 23 months after resection of a metastatic esophageal lesion from primary lung cancer (18). The patient in our case received chemotherapy and apatinib for molecular targeted therapy without surgery, due to multiple enlarged lymph nodes in the retroperitoneum and pelvic cavity.
During chemotherapy, the patient had disease progression within 6 months of being diagnosed. Considering patient’s medical economic burden, we commenced targeted treatment with apatinib orally instead of expensive bevacizumab. Unexpectedly, stable disease was maintained for 6 months after administration of apatinib and has continued to be maintained. Apatinib is a small-molecule antiangiogenic agent that selectively inhibits vascular endothelial growth factor receptor-2 (VEGFR-2). Apatinib has been studied and developed in China and is recommended for anti-tumor activity against a wide range of tumors, including gastric carcinoma, breast carcinoma, non-small cell lung carcinoma, hepatocarcinoma, and soft tissue tumors (19). It was proved and lunched by China in 2014 as a subsequent-line treatment for patients with advanced gastric cancer or gastroesophageal junction adenocarcinoma. In a randomized, placebo-controlled, double-blind phase III trial (NCT01512745), Qin et al. found that advanced gastric cancer patients who previously failed to second-line chemotherapy had significantly prolonged median progression-free survival (mPFS) (78 vs. 53 days) compared with those treated with placebo (20). In addition, it is also currently undergoing phase II/III clinical trials in China for the treatment of many cancer types, such as non-small cell lung cancer (NSCLC), breast cancer, and hepatocellular carcinoma. Bevacizumab, a monoclonal antibody directed against vascular endothelial growth factor (VEGF), was found to effectively control multiple solid tumors, including recurrent metastatic colorectal cancer, recurrent non-squamous NSCLC, progressive glioblastoma, metastatic renal cell carcinoma, platinum-resistant and platinum-sensitive recurrent epithelial ovarian cancer, metastatic cervical cancer, fallopian tube cancer, and primary peritoneal cancer (21-23). Bevacizumab and apatinib mainly differ with respect to their drug target. Moreover, patients treated with apatinib spend only half of money compared with bevacizumab in china. This case is the first report of significant activity of the single agent apatinib in a patient with metastatic esophageal carcinoma from endometrial adenocarcinoma, which was not responsive to chemotherapy. The patient’s mPFS had been prolonged for 3 months. We believe that our report suggests a new way to treat this disease.
In summary, this is the second case that presented a case of secondary esophageal carcinoma derived from an endometrial adenocarcinoma. Based on the results of clinical examination and immunohistochemical staining, the diagnosis of endometrial origin was demonstrated. Apatinib may be a promising new therapeutic option for these patients. However, further investigations are warranted to determine the optimal targeted agent for this disease.
Acknowledgments
Funding: The present Case Report was supported by grants from Zhejiang Provincial Medical Hygienic Science and Technology Project of China (No. 2013KYA104) and Education Department of Zhejiang Province (No. Y201534639).
Footnote
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/tcr.2018.06.08). The authors have no conflicts of interest to declare.
Ethical statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. All procedures performed in study involving human participants were in accordance with the ethical standards of the institutional and/or national research committee(s) and with the Declaration of Helsinki (as revised in 2013). Written informed consent was obtained from the patient for publication of this case report and any accompanying images.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Abrams HL, Spiro R, Goldstein N. Metastases in carcinoma. Analysis of 1000 autopsied cases. Cancer 1950;3:74-85. [Crossref] [PubMed]
- Mizobuchi S, Tachimori Y, Kato H, et al. Metastatic esophageal tumors from distant primary lesions: report of three esophagectomies and study of 1835 autopsy cases. Jpn J Clin Oncol 1997;27:410-4. [PubMed]
- Agha FP. Secondary neoplasms of the esophagus. Gastrointest Radiol 1987;12:187-93. [PubMed]
- Fisher MS. Metastasis to the esophagus. Gastrointest Radiol 1976;1:249-51. [PubMed]
- Murakami T, Elquza E, Ozden N, et al. A Rare finding of direct esophageal metastasis from a pancreatic primary tumor. Pancreas 2014;43:1123-5. [Crossref] [PubMed]
- Hsu P-K, Shai S-E, Wang J, et al. Esophageal metastasis from occult lung cancer. J Chin Med Assoc 2010;73:327-30. [Crossref] [PubMed]
- Fukatsu H, Miura S, Kishida H, et al. Gastrointestinal: Esophageal metastasis from hepatocellular carcinoma. J Gastroenterol Hepatol 2012;27:1536. [Crossref] [PubMed]
- Gross P, Freedman LJ. Obstructing secondary carcinoma of the esophagus. Arch Pathol Lab Med 1942;33:361-4.
- Simchuk E, Low D. Direct esophageal metastasis from a distant primary tumor is a submucosal process: a review of six cases. Dis Esophagus 2001;14:247-50. [Crossref] [PubMed]
- Zarian LP, Berliner L, Redmond P. Metastatic endometrial carcinoma to the esophagus. Am J Gastroenterol 1983;78:9-11. [PubMed]
- Lagendijk J, Mullink H, Van Diest P, et al. Immunohistochemical differentiation between primary adenocarcinomas of the ovary and ovarian metastases of colonic and breast origin. Comparison between a statistical and an intuitive approach. J Clin Pathol 1999;52:283-90. [Crossref] [PubMed]
- Arif A, Abideen ZU, Zia N, et al. Metastatic involvement of the spleen by endometrial adenocarcioma; a rare asylum for a common malignancy: a case report. BMC Res Notes 2013;6:476. [Crossref] [PubMed]
- Mittal K, Soslow R, McCluggage W. Application of immunohistochemistry to gynecologic pathology. Arch Pathol Lab Med 2008;132:402-23. [PubMed]
- Wang NP, Zee S, Zarbo RJ, et al. Coordinate expression of cytokeratins 7 and 20 defines unique subsets of carcinomas. Appl Immunohistochem 1995;3:99-107.
- Liang L, Zheng W, Liu J, et al. Assessment of the utility of PAX8 immunohistochemical stain in diagnosing endocervical glandular lesions. Arch Pathol Lab Med 2016;140:148-52. [Crossref] [PubMed]
- Atkins JP. XXVII Metastatic Carcinoma to the Esophagus: Endoscopic Considerations with Special Reference to Carcinoma of the Breast. Ann Otol Rhinol Laryngol 1966;75:356-67. [Crossref] [PubMed]
- Shimada Y, Imamura M, Tobe T. Successful esophagectomy for metastatic carcinoma of the esophagus from breast cancer--a case report. Jpn J Surg 1989;19:82-5. [Crossref] [PubMed]
- Oka T, Ayabe H, Kawahara K, et al. Esophagectomy for metastatic carcinoma of the esophagus from lung cancer. Cancer 1993;71:2958-61. [Crossref] [PubMed]
- Zhang H. Apatinib for molecular targeted therapy in tumor. Drug Des Devel Ther 2015;9:6075. [Crossref] [PubMed]
- Qin S. Phase III study of apatinib in advanced gastric cancer: a randomized, double-blind, placebo-controlled trial. J Clin Oncol. 2014;32:15_suppl; abstr 4003.
- Minion LE, Tewari KS. The safety and efficacy of bevacizumab in the treatment of patients with recurrent or metastatic cervical cancer. Expert Rev Anticancer Ther 2017;17:191-8. [Crossref] [PubMed]
- Bidus MA, Webb JC, Seidman JD, et al. Sustained response to bevacizumab in refractory well-differentiated ovarian neoplasms. Gynecol Oncol 2006;102:5-7. [Crossref] [PubMed]
- Sugiyama T, Mizuno M, Aoki Y, et al. A single-arm study evaluating bevacizumab, cisplatin, and paclitaxel followed by single-agent bevacizumab in Japanese patients with advanced cervical cancer. Jpn J Clin Oncol 2017;47:39-46. [Crossref] [PubMed]


