
A case report of primary anaplastic large cell lymphoma arising from the trachea
Introduction
Primary malignant tumors of the trachea are rare, accounting for about 0.1–0.4% of all malignancies (1-3). The most common pathology is squamous cell carcinoma, followed by cystadenoma (Table 1) (1,4). Although extranodal lymphoma is commonly encountered, lymphoma presenting in the large airways are incredibly rare (5), constituting less than 3% of tracheal tumors. Less than 10 cases have been reported in the literature to date (6,7). Presenting symptoms can be mistaken for other entities, and imaging can play a vital role in diagnosis and management. Here, we present a case of anaplastic large cell lymphoma (ALCL) of the trachea with an emphasis on the imaging features of this entity and a discussion of treatment complications.
Table 1
| Features | Adenoid cystic carcinoma | Carcinoid | Mucoepidermoid carcinoma | Lymphoma |
|---|---|---|---|---|
| Predilection site | Trachea or main bronchus; the main body is in the lateral posterior wall | Main bronchus | Lobar or segmental bronchus, rare in trachea or main bronchus | Trachea or main bronchus |
| Growth characteristic | Mass encircling the trachea with wall thickening; usually involve more than 180° of the airways circumferences; both intra and extra the lumen | Nodule within the lumen and mass in the external lumen | Smooth circular or round like nodule attached the wall, with broad base. The long axis is parallel to the lumen | Mass within the tracheobronchial lumen |
| Density | Homogeneous | Homogeneous; flake necrosis can be seen in atypical type | Mostly homogeneous; lower than muscle | Homogeneous; necrosis is rare |
| Calcification | Rare | Characteristic manifestation: diffuse, eccentric; gravel, nodular, popcorn | Punctate or nodular | Rare |
| Enhancement | Mild | Obvious | Mild heterogeneous enhancement | Mild homogeneous enhancement |
Case presentation
A 31-year-old man presented to our institution complaining of a 3-month history of productive cough and 1 week of shortness of breath. The cough was associated with chest pain and hemoptysis. The patient denied fevers, fatigue or loss of appetite. A physical exam was notable in that the patient exhibited a high-pitched wheeze in the throat and upper chest.
High resolution computed tomography (CT) was performed, which demonstrated a 2.0 cm × 2.0 cm × 1.6 cm endotracheal nodule extending from the lower right tracheal wall (Figure 1). The lesion was homogeneous with mild contrast enhancement. It caused near-complete occlusion of the trachea. In addition, there was a right paratracheal lesion adjacent to the endotracheal mass with extension into the medial right upper lobe.
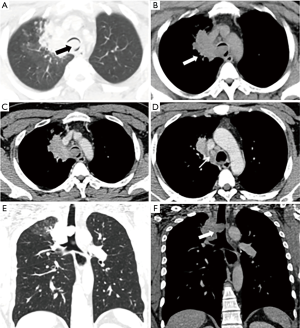
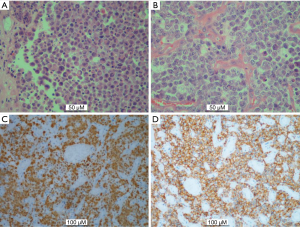
Resection of the tracheal lesion and sampling of the right paratracheal mass was performed via bronchofiberoscopy. Histology was consistent with ALCL (Figure 2). Anaplastic lymphoma kinase (ALK) staining was positive. Other immunohistochemistry stains included: CD30(+), CD8(−), CD4(−), CD5(−), Granzyme B(+), Perforin(+), TIA-1(+), CD2(−), CD43(−), CD68(−), EMA(+), PLAP(−), Myogenin(−), Des(−), MyoD1(weak +), CD99(+), Myoglobin(+), Vim(+), LCA(+), and Ki67(90%+).
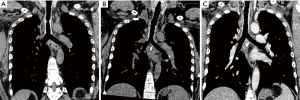
A bronchial stent was subsequently placed after mass resection, followed by autologous stem cell transplantation, chemotherapy and radiotherapy (Figure 3). Patient’s dyspnea markedly improved post-operatively. The right paratracheal mass also significantly decreased in size. The patient was subsequently admitted multiple times over the course of 4 years for recurring anhelation. CT imaging from his first post-operative hospitalization raised concern for tumor recurrence, given the increased tracheal wall thickening that resulted in re-stenosis (Figure 4). Trans-bronchial biopsy performed at that time demonstrated granulomatous changes without evidence of lymphoma. This tissue was resected, and the stent exchanged. The patient has required further stent exchanges due to recurrence of granulomatous tissue, but no evidence of tumor recurrence has been noted to date.
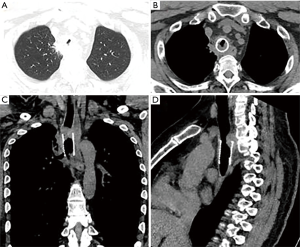
Discussion
ALCL is a sub-type of non-Hodgkin lymphoma (NHL) (8) and can be categorized as ALK-positive and ALK negative. The distinction of them is important and essential because of clinical and prognostic differences. Namely, ALK-positive ALCL is more common in younger patients who generally have superior outcomes when treated with standard chemotherapy as compared to patients with ALK-negative ALCL. ALK-positive ALCL patients typically have 5-year overall survival rates of more than 70%, whereas patients with ALK-negative ALCL have less than 50% (8,9). Those with ALK-negative ALCL are often middle-aged patients (8). There is a strong (6:1) male predilection.
Clinical manifestations of tracheal ALCL, including dyspnea, stridor and wheezing due to obstruction (8,10), can rapidly progress to respiratory failure and require emergency management. They can often be initially misdiagnosed as chronic obstructive pulmonary disease. Thus, if the above symptoms are present, it is important to consider the presence of a large airway tumor on the differential diagnosis.
Airway lesions are difficult to diagnose on chest radiography and CT is very helpful for imaging evaluation. ALCL mainly occur in the trachea or main bronchus, manifesting as a mass within the tracheobronchial lumen. Pharyngeal lymphoma does occur but is also a rare entity (11). These masses are generally not necrotic, unlike other tracheal lesions (Table 1). Most of the cases present locally, but involvement of the adjacent mediastinum and lung can sometimes occur. Because of patients’ airway symptoms, patients are usually diagnosed prior to significant mediastinal or parenchymal involvement. Diffuse lymphadenopathy is not common in primary tracheal ALCL.
Treatment strategies for patients with primary tracheal lymphoma are controversial and may depend on pathology subtype. Surgery, chemotherapy, and radiation therapy usually are used either alone or in combination. Positron emission tomography (PET)/CT maybe useful to search for systemic disease and for treatment response evaluation. The International Prognostic Index (IPI) is important for evaluating NHL, which was determined using prognostic factors (the age at diagnosis, performance status, lactate dehydrogenase (LDH), stage and number of extranodal sites). This patient had a low-risk IPI, with 0 risk factors. For patients whose LDH was unknown, it was presumed to be normal for calculating the IPI.
Local resection is usually performed for symptom relief. Relapsing disease is very uncommon, but has been known to occur. Thus, imaging follow-up is important in these patients. Even without relapse, complications from local benign granulomatous growths can lead to recurrent clinical symptoms (12). For our patient, the increased soft tissue around the endobronchial stent required multiple debridement and stent exchanges to maintain airway patency. Quantitative evaluation of the airway prior to stent placement, which can be provided by imaging, may be useful to reduce this complication.
In conclusion, primary ALCL is a rare endotracheal tumor with presenting symptoms that can often be misdiagnosed as chronic obstructive pulmonary disease. Taken together with patient’s clinical history, imaging features on CT can help guide the clinician to consider this diagnosis. Follow-up imaging post treatment is important to monitor treatment complications and, in rare cases, disease recurrence.
Acknowledgments
Finding: The work of H Chen and S Huang were supported by
Footnote
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/tcr.2019.02.05). The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee(s) and with the Helsinki Declaration (as revised in 2013). Written informed consent was obtained from the patient for publication of this manuscript and any accompanying images.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Khodadad K, Karimi S, Arab M, et al. Primary anaplastic large cell lymphoma of trachea with subcutaneous emphysema and progressive dyspnea. Hematol Oncol Stem Cell Ther 2011;4:188-91. [Crossref] [PubMed]
- Mizuno S, Ota S, Tanaka T, et al. Primary tracheal malignant lymphoma detected during a regular checkup in an asbestos dust-exposed smoker. Acta Med Okayama 2014;68:177-81. [PubMed]
- Xu X. ALK-negative anaplastic large cell lymphoma primarily involving the bronchus: a case report and literature review. Int J Clin Exp Pathol 2013;7:460-3. [PubMed]
- Chand T, Bansal A, Dua H, et al. Endotracheobronchial lymphoma: Two unusual case reports and review of article. Lung India 2016;33:653-6. [Crossref] [PubMed]
- Wang AY, Wang JT, Shen Y, et al. Relapse of Non-Hodgkin's Lymphoma Involving the Trachea: Acute Subglottic Obstruction. Case Rep Otolaryngol 2014;2014:230682. [Crossref] [PubMed]
- Park HS, McIntosh L, Braschi-Amirfarzan M, et al. T-Cell Non-Hodgkin Lymphomas: Spectrum of Disease and the Role of Imaging in the Management of Common Subtypes. Korean J Radiol 2017;18:71-83. [Crossref] [PubMed]
- Zhao Q, Liu Y, Chen H, et al. Successful Chemo-Radiotherapy for Primary Anaplastic Large Cell Lymphoma of the Lung: A Case Report and Literature Review. Am J Case Rep 2016;17:70-5. [Crossref] [PubMed]
- Fidias P, Wright C, Harris NL, et al. Primary tracheal non-Hodgkin's lymphoma. A case report and review of the literature. Cancer 1996;77:2332-8. [Crossref] [PubMed]
- Dong X, Li J, Huo N, et al. Primary central nervous system ALK-positive anaplastic large cell lymphoma in an adult: A rare case report. Medicine (Baltimore) 2016;95:e5534. [Crossref] [PubMed]
- Fernandez FG, Denlinger CE, Crabtree TD. Primary non-Hodgkin's lymphoma of the trachea. J Thorac Oncol 2010;5:403-4. [Crossref] [PubMed]
- Siddiqui NA. Imaging characteristics of primary laryngeal lymphoma. AJNR Am J Neuroradiol 2010;31:1261-5. [Crossref] [PubMed]
- Hu HC, Liu YH, Wu YC, et al. Granulation tissue formation following Dumon airway stenting: the influence of stent diameter. Thorac Cardiovasc Surg 2011;59:163-8. [Crossref] [PubMed]


