
Initial clinical experience with surface image guided (SIG) radiosurgery for trigeminal neuralgia
Introduction
Trigeminal neuralgia (TN) is a disorder of the cranial nerve that is characterized by extreme, shock-like pain in areas of the face where the trigeminal nerve is distributed. Patients suffering from TN commonly experience episodes of pain when doing activities such as brushing teeth, combing hair, talking, chewing, or touching the face. According to the National Institute of Neurological Disorders and Stroke, the incidence of new cases is approximately 12 per 100,000 people per year (1). TN is thought to be most commonly caused by an enlarged blood vessel, usually the superior cerebellar artery, pressing against the trigeminal nerve and damaging the myelin sheath coating the nerve.
There are numerous treatments for TN, such as anticonvulsant medications, percutaneous rhizotomies, and microvascular decompression (MVD). Increasingly patients who do not tolerate medication side effects or who do not obtain durable pain relief from medications alone, and do not wish to undergo invasive surgeries are turning to stereotactic radiosurgery. Stereotactic radiosurgery is a form of treatment in which highly focused beams of radiation are directed at the site where the trigeminal nerve exits the brain stem. Following irradiation, a lesion slowly forms and disrupts the pain fibers of the trigeminal nerve (1,2). Various published reports have found that 59-94% of patients experienced some level of facial pain relief (3-5).
The success of stereotactic treatment is highly dependent on accurate and stable patient immobilization. The largest published experiences of SRS for TN utilized Gamma Knife (3-6). Two common methods used to immobilize patients for SRS with Gamma Knife are the Brown-Roberts-Wells (BRW) frame and the Gill-Thomas-Cosman (GTC) frame (6). However, the BRW frame is invasive and the GTC frame can be slightly inaccurate (7); both are uncomfortable for patients. An alternative is what was originally termed frame-less, mask-less, and bite block-less (FMB) stereotactic radiosurgery. Reports using this same technique were later changed to surface image guided radiosurgery (SIG-RS) (8). With this method of immobilization, an open-faced mask (CIVCO, Medical Solutions, Kalona, Iowa) with added rigidity around the forehead and mandible is paired with a custom SRS headrest (AccuForm, CIVCO Medical Solutions). The face is left completely uncovered allowing the facial landmarks to be monitored by the surface imaging system while promoting patient comfort and convenience (Figure 1). Surface image guided immobilization uses an imaging system that tracks the topography of the patient’s face as a surrogate for the isocenter, and automatically corrects for positioning errors. The SIG-RS arrangement has been shown to be highly accurate when employed with a real-time surface imaging system (9). We report the methodological details of our SIG-RS setup as well as our initially clinical experience for seven patients treated for TN.
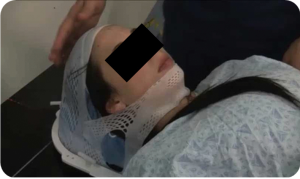
Methods
Patients underwent stereotactic-protocol T1-weighted brain MRI with contrast enhancement and T2-weighted volume acquisition divided into 1 mm slices. At the time of CT simulation, patients were setup in a supine position on a pad with a knee roll, and a headrest placed under their neck on a Type-S board (CIVCO Medical Solutions, Kalona, IA). Patients were positioned such that the sagittal laser intersects with the midline of the brain, and an AccuForm cushion (CIVCO Medical Solutions, Kalona, IA) was custom-molded between the patient’s neck and the headrest. A thermoplastic open faced mask (CIVCO Medical Solutions, Kalona, IA) was placed on the patient’s head and attached to the Type-S board. Patients underwent simulation with a non-contrast CT (1.25 mm slice thickness) prior to the procedure using the respective immobilization devices.
Following simulation, the MRI and CT images were registered using the rigid registration module in Eclipse treatment planning system, version 8 (Varian Medical Systems, Palo Alto, CA) and the neurosurgeon and radiation oncologist jointly contoured the trigeminal nerve root. The CT-generated body contour was auto-contoured using a Hounsfeld unit (HU) cutoff of –350 HU. Brainstem, optic chiasm, optic nerves, brain, eyes and eye lenses were contoured. Planning was done with the cone planning module in Eclipse and normalization was to the 80% isodose line. Each plan consisted of 13 noncoplanar arcs using 5 mm cones (Figure 2). The CT body contour and plan were exported to the optical surface imaging system, AlignRT system (Vision RT Ltd., London, UK). At the surface imaging console, the contour and plan were imported, and a region of interest (ROI) was selected to designate the section of the surface contour to be used for surface monitoring (Figure 3). When selecting the ROI, the eyes and tip of nose were excluded.
On the day of treatment, the S-frame was placed on the treatment couch and the proper cone was inserted on the gantry accessory mount. Patients were setup on the couch with the knee fix and head rest prior to placing the open-faced mask over their head and attaching it to the S-frame. Patient initial setup and intrafraction motion monitoring was performed using the AlignRT system. The AlignRT system consisted of three ceiling-mounted camera pods (two lateral and one at the foot of the treatment couch). Three camera pods were used to ensure that at least two pods could view the patient surface at all gantry and couch angles. Each AlignRT pod has two cameras used for stereovision, a speckle projector that projects a unique speckle pattern, and a texture camera. The two cameras read the pseudorandom speckle pattern and reconstruct the data as a 3-dimensional surface using photogrammetry. The translational and rotational disparities between the CT-generated surface and the treatment surface are displayed in real time. Translational errors are specified for lateral, longitudinal, vertical, and the vector sum. Rotational errors are specified as yaw, pitch, and roll. During initial setup, the patient position is fine-tuned until the translational and rotational disparities are all less than 1 mm and 1°, respectively.
After setting the patient up with AlignRT, a right-lateral and anterior-posterior kV image was acquired with the on board imaging system. The planar images were registered to the digitally reconstructed radiographs (DRRs) from the planning CT, and rotational discrepancies were fixed, if necessary. A full-fan cone beam CT was acquired with 1 mm slices, registered to the planning CT, and reviewed by a radiation oncologist and physicist. After registration, the necessary couch shifts were applied, and a new reference surface image was acquired by AlignRT to be used for intrafraction movement monitoring. Once the new reference surface image was acquired, the translational and rotational deltas were monitored to ensure that the ROI was within 1 mm and 1°. If the patient moved enough to exceed these tolerances, the beam was turned off and the patient was readjusted to be within tolerance prior to continuing treatment.
Following treatment, patients were initially seen 4-6 weeks later and then again every 6 months or as needed to evaluate for response and treatment complications.
Results
Seven patients with TN with either a BNI pain score of (IV) (pain, not well controlled with medication) or (V) (pain without any relief from medication) were treated with SIG-RS (Table 1). All patents received treatment in a single fraction with dose ranging from 80-90 Gy. Following treatment, all patients experienced pain relief. The BNI pain score of four of the seven patients following treatment was (I) which equates to complete resolution of pain. The remaining three patients had pain consistent with BNI of (II) which equates occasional pain not requiring medication for control. Two of the seven patients developed recurrences of pain during a median follow up across all seven patients of 31.4 months. Both patients’ pain returned as BNI (IV), one at 52 months and the other at 31 months after treatment. One patient reported intermittent facial numbness which resolved 9 months following treatment.
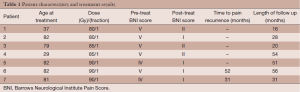
Full table
For the patients presented in this series, the planned brainstem point max ranged from 8.97 to 12 Gy. The IMRT factor—ratio of monitor units to prescription dose in cGy—ranged from 2.21 to 2.34 for the plans using 13 noncoplanar arcs with 5 mm cones, and the total monitor units ranged from 18,116 to 21,085 MU.
Discussion
This article reviews our early clinical experience with SIG-RS for TN. Full details of patient setup and monitoring have been previously described by Cerviño and colleagues (9) and updated by Pan et al. using the thermoplastic open faced mask (CIVCO medical solutions, Kalona, IA) (8). With SIG-RS, precise immobilization is achieved while providing improved patient cooperation and comfort. Patient motion is monitored using a non-ionizing surface imaging system providing initial setup verification and intra-fraction tracking. The system allows the beam delivery to be stopped if motion exceeds a pre-specified tolerance. The correct position of the patient is always prioritized, and, therefore, the accuracy of the treatment is not to be compromised in cases of prolonged treatment times. In our practice, initial setup by the surface imaging system is always confirmed and finalized with cone-beam CT (CBCT) imaging, which continues to be the gold standard for position verification.
Interest in radiosurgery utilizing a SIG technique is increasing as it has been demonstrated to achieve maximum patient comfort by obviating the need for rigid immobilization (8,9). A potential concern with open face immobilization using the thermoplastic mask described in this study is the potential risk for poorer localization when compared to traditional frame-based or other mask-based systems. In our previously published series, we have shown that this technique provides excellent immobilization while maintaining patient comfort (8,9). However, we fully appreciate that for certain patients with circumstances that may prevent them from fully engaging and understanding instructions regarding setup and maintenance of the same position through treatment time, more traditional treatment techniques such as mask- or frame-based techniques might be more appropriate.
In our clinic, the implementation of the SIG-RS has been performed with the assumption that the motion of the patient’s monitored surface and the motion of the target are perfectly correlated; that is, that the target moves if and only if patient’s monitored surface moves. Changes in facial expression, or even blinking, could potentially lead to the surface imaging system inaccurately reflecting target motion. In a previous study, the surface imaging system showed very small motion during the volunteers’ 20-minute monitoring time, proving for these cases that volunteers did not change facial expression, or that facial expressions had very little or no effect on monitoring results (10). In addition, we choose regions of interest on the face to monitor that have limited to no motion (forehead, zygomatic arch). Another strength of our current treatment protocol is that position is also verified with CBCT imaging to assess surface-image guidance correlation.
In summary, we report our experience using the SIG-RS procedure which features open-face thermoplastic mask immobilization and precise patient monitoring with a surface imaging system for single-fraction radiosurgery for TN. This technology is a significant leap forward in patient comfort and treatment time when compared to more commonly use framed and frameless-based stereotactic techniques. The emergence and application of precise surface imaging technology has made SIG-RS safe and appropriate for even single fraction, high dose treatments. While this is a small initial series using SIG-RS for TN, our treatment results are thus far comparative to traditional frame-based systems (11-13). The excellent pain relief and lack of treatment complications in this cohort is evidence not only of the safety of this technique but also of its excellent target localization.
Acknowledgments
Funding: None.
Footnote
Provenance and Peer Review: This article was commissioned by the editorial office, Translational Cancer Research for the series “SBRT/SRS in Radiation Research”. The article has undergone external peer review.
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.3978/j.issn.2218-676X.2014.07.03). The series “SBRT/SRS in Radiation Research” was commissioned by the editorial office without any funding or sponsorship. KTM served as the unpaid Guest Editor of the series and serves as an unpaid editorial board member of Translational Cancer Research. The authors have no other conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. The study was conducted in accordance with the Declaration of Helsinki (as revised in 2013). The study was approved by the institutional ethical committee. Written informed consent was obtained from the patients.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Kitt CA, Gruber K, Davis M, et al. Trigeminal neuralgia: opportunities for research and treatment. Pain 2000;85:3-7. [PubMed]
- Baschnagel AM, Cartier JL, Dreyer J, et al. Trigeminal neuralgia pain relief after gamma knife stereotactic radiosurgery. Clin Neurol Neurosurg 2014;117:107-11. [PubMed]
- Dhople AA, Adams JR, Maggio WW, et al. Long-term outcomes of Gamma Knife radiosurgery for classic trigeminal neuralgia: implications of treatment and critical review of the literature. Clinical article. J Neurosurg 2009;111:351-8. [PubMed]
- Elaimy AL, Lamm AF, Demakas JJ, et al. Gamma knife radiosurgery for typical trigeminal neuralgia: An institutional review of 108 patients. Surg Neurol Int 2013;4:92. [PubMed]
- Song ZX, Qian W, Wu YQ, et al. Effect of the gamma knife treatment on the trigeminal nerve root in Chinese patients with primary trigeminal neuralgia. Turk Neurosurg 2014;24:163-9. [PubMed]
- Verbakel WF, Lagerwaard FJ, Verduin AJ, et al. The accuracy of frameless stereotactic intracranial radiosurgery. Radiother Oncol 2010;97:390-4. [PubMed]
- Das S, Isiah R, Rajesh B, et al. Accuracy of relocation, evaluation of geometric uncertainties and clinical target volume (CTV) to planning target volume (PTV) margin in fractionated stereotactic radiotherapy for intracranial tumors using relocatable Gill-Thomas-Cosman (GTC) frame. J Appl Clin Med Phys 2010;12:3260. [PubMed]
- Pan H, Cerviño LI, Pawlicki T, et al. Frameless, real-time, surface imaging-guided radiosurgery: clinical outcomes for brain metastases. Neurosurgery 2012;71:844-51. [PubMed]
- Cerviño LI, Detorie N, Taylor M, et al. Initial clinical experience with a frameless and maskless stereotactic radiosurgery treatment. Pract Radiat Oncol 2012;2:54-62. [PubMed]
- Peng JL, Kahler D, Li JG, et al. Characterization of a real-time surface image-guided stereotactic positioning system. Med Phys 2010;37:5421-33. [PubMed]
- Adler JR Jr, Bower R, Gupta G, et al. Nonisocentric radiosurgical rhizotomy for trigeminal neuralgia. Neurosurgery 2009;64:A84-90. [PubMed]
- Sheehan J, Pan HC, Stroila M, et al. Gamma knife surgery for trigeminal neuralgia: outcomes and prognostic factors. J Neurosurg 2005;102:434-41. [PubMed]
- Kondziolka D, Zorro O, Lobato-Polo J, et al. Gamma Knife stereotactic radiosurgery for idiopathic trigeminal neuralgia. J Neurosurg 2010;112:758-65. [PubMed]




