
Analysis of a pedigree of Peutz-Jeghers syndrome and RET proto-oncogene mutation: one case report and literature review
Introduction
Peutz-Jeghers syndrome (PJS) is characterized by gastrointestinal hamartomas. The incidence has been estimated between 1:8,300 and 1:200,000 births, and no significant gender or racial differences were found (1). There’s been a study demonstrates that PJS patients carry a high cumulative risk of intussusception already early in life, the intussusception risk is 50% at age 20, the intussusceptions occur in the small intestine in 95% of events, while treatment is mostly surgical (2). The hamartomas are located predominantly in the small intestine and colon, which may cause intussusceptions. The hamartomas are located in the small intestine in >90% of patients, predominantly in the jejunum (3). Even in the early childhood, the hamartomas may induce complications including bleeding, abdominal pain, anemia, and intussusception (4). In this study, we took PJS pedigree as the research object, the first symptom of the proband was intussusception. We collected the clinical data after a follow-up of 8 years. Through a new generation of Sanger sequencing technology, we found that there were three heterozygous mutations of RET gene. This pedigree investigation showed that it is critical to establish a long-term follow-up system for PJS patients and their families.
Object and methods
Object of study
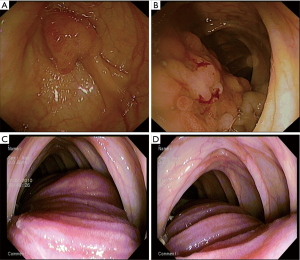
The female proband, aged 34 years old, was found to have dark spots in the mucous membrane of lips and cheeks, as well as plantar pigmentation at age 3. On January 5, 2010, the patient visited the outpatient department of our hospital due to abdominal pain. The results of enteroscopy suggested multiple polyps of colon. On January 15, 2010, the patient was admitted in the Digestive Department of our hospital because of intermittent abdominal pain lasted for 4 years and loss of appetite for half a year. Laboratory analysis of blood showed that the patient had anemia. After admission, the patient was desired to accept endoscopic polypectomy on January 21, 2010, and findings during the inspection revealed that multiple polyps of the colon (Figure 1A,B), meanwhile longitudinal structure of small intestine was visible in the ascending colon that appeared cyanotic, which was considered to be intussusception (Figure 1C). Endoscopic reversion of colon was tried, but failed; meanwhile, manual reduction of intussusception, partial resection of small intestine and excision of intestinal polyp were performed on the same day. Intraoperative findings: multiple polyps of small intestine, resulting in the occurrence of intussusception in five sites, and the most severe site was at the terminal ileum, about 1m of the ileum was inserted into the ascending colon. Following manual reduction, a polyp in a size of about 3.0 cm × 2.0 cm was found at 20 cm away from the ileocecus, which was pathologically examined to be hamartomatous polyp postoperatively (Figure 1D). The patient was discharged on February 01, 2010 with successful postoperative outcome. Subsequently, in 2010, 2011, 2012 and 2013, the patient underwent multiple high-frequency electrical resection of polyps under endoscopy and colonoscopy, and argon plasma coagulation therapy, respectively. On December 01, 2013, the patient was admitted to the Department of General Surgery of our hospital due to the discovery of right thyroid mass for 1 year and received excision of right thyroid tumor. Postoperative pathological findings: nodular goiter, and cystic degeneration. After that, review of colonoscopy and gastroscopy, as well as concurrent endoscopic polypectomy were performed every year in 2014, 2015,2016 and2017 respectively.
Family history
The parents of the proband were in good condition, and the sister had dark spots of lips who had died of intussusception at the age of 18. The older daughter was aged 11 and had been found to have dark spots in the mucous membrane of lips and cheeks at age 2. Gastroscopy and colonoscopy have been performed and no polypoid lesions were found. The younger daughter was 6 years old without the occurrence of skin pigmentation. Intermittent diarrhea and abdominal pain occurred in the past year (there is still no endoscopic information).
Methods and results
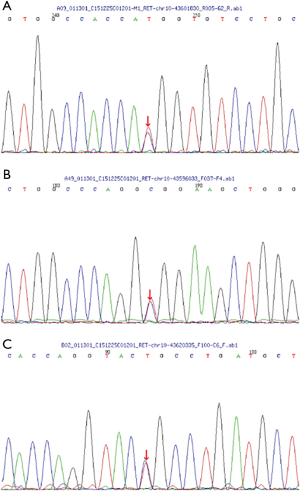
During the annual follow-up process, the mother of the family underwent gastrointestinal microscopic examination and endoscopic polyp therapy. Through a new generation of gene capture sequencing, the mother and two daughters both detected three heterozygosity mutations in the RET gene, both from the mother and the father as a family health control did not find the mutation. The mutation site is as follows: chr10-43601830, chr10-43596033, chr10-43620335, corresponding nucleotide changes: c.G874A, c.G200A, c.C2944T (Figure 2).
Discussion
PJS, described successively by Peutz (in 1921) (5) and Jeghers (in 1949) (6), is a rare autosomal dominant disease characterized by hyperpigmentation in the mucous membranes of the skin and multiple polyps of the gastrointestinal tract.
Pigmentation of the skin is one of the characteristic clinical manifestations of the patient. It usually occurs on the lips and lips, skin, cheeks, mucous membranes, fingers and toes, meanwhile, pigmentation of the nasal, perianal, genital, and orbital regions can also be observed. Pigmentation often occurs at birth or in infancy, and as the age increases, the dark spot of a part of the patient can disappear gradually. The mother and daughters of the family were seen with dark spots in their mucous membranes of lips and cheeks, as well as in fingers. Both patients presented skin and mucous membrane pigmentation at the younger age, which was consistent with the clinical characteristics of PJS.
PJS characterized polyps are mainly gastrointestinal hamartoma polyps, which can be distributed throughout the digestive tract, most commonly in the small intestine (65%), followed by the colon (50%), stomach (50%), rectum (30%), and are also found in the upper and lower respiratory tract and urinary bladder (7). Polyps are usually multiple, with different size and shape, easy to lead to intestinal obstruction and intussusception, increased rate of emergency operation, and possessing a malignant tendency, thereby resulting in a decreased quality of life of patients. In this case, the mother was desired to receive treatment for colonic polyp at her first visit, and intussusception was found during enteroscopy. On the same day, the patient received manual reduction of intussusception, partial resection of small intestine and excision of intestinal polyp. Multiple polyps of the small intestine were seen during the operation. A survey of the pedigree found that the sister of the proband died of intussusception, which was similar to the result of the study performed by van Lier et al. that the risk of intussusception in PJS patients was about 50% at age 20, and long-term colonoscopy monitoring might be beneficial for reducing the risk of intussusception.
During the following 8 years of follow-up, review of colonoscopy and gastroscopy, as well as concurrent endoscopic polypectomy were performed every year, and no malignant polyps were found for the moment. Furthermore, the older daughter had underwent gastrointestinal endoscopy, and no polypoid lesions of the stomach or colon were detected.
It is reported that mutations in STK11 gene can be found in 70–80% PJS patients (8). However, PJS has genetic heterogeneity, and STK11 mutation cannot be detected in all PJS patients. There was no discovery of a specific pathogenic mutation of the STK11 gene in our follow-up study. Through Sanger sequencing and bioinformatics analysis, three heterozygous mutations were revealed in the RET gene that were inherited from the mother, all of which were missense mutations caused by base substitutions, and no mutation was found in their father, a healthy control within the family. RET is located on the long arm of chromosome 10 and encodes a transmembrane protein belonging to the superfamily of tyrosine kinase receptors. RET gene mutation is considered to be the major causative factor and genetic basis of multiple endocrine neoplasia type 2 (MEN2). Current research result documented that mutations in the RET gene were associated with congenital megacolon and medullary thyroid carcinoma (9).
In our research, the studied family members, including the proband, older daughter and younger daughter, all detected to have three heterozygous mutations in the RET gene that were inherited from the proband. The existed three mutant spots included exon 5 (GTG>ATG, Val292Met), exon 2 (CGC>CAC, Arg67His) and exon 18 (CGC>TGC, Arg982Cys) in RET. Our study provides an observation of the genetic heterogeneity of PJS. However, the disadvantage is that the target genes for hereditary tumor are used to capture the sequencing instead of whole genome sequencing, which may lead to the missing of some information. Significantly, more PJS pedigrees and sporadic cases should be confirmed for the relationship between RET gene mutations and the pathogenesis of PJS.
Therefore, genetic sequencing and genetic diagnosis of PJS family members are useful in screening high-risk group within the family, and mostly importantly, it will contribute to the establishment of a long-term endoscopic follow-up system for PJS patients and family members to pay attention to the possibility of malignant transformation in polyps, as well as other gastrointestinal and non-gastrointestinal tumors.
Acknowledgments
Funding: None.
Footnote
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/tcr.2020.02.64). The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. Consent was obtained from relative of the patient for publication of this report and any accompanying images. All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee(s) and with the Helsinki Declaration (as revised in 2013).
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Sengupta S, Bose S. Peutz-Jeghers Syndrome. N Engl J Med 2019;380:472. [Crossref] [PubMed]
- van Lier MG, Mathus-Vliegen EM, Wagner A, et al. High cumulative risk of intussusception in patients with Peutz-Jeghers syndrome: time to update surveillance guidelines? Am J Gastroenterol 2011;106:940-5. [Crossref] [PubMed]
- McGarrity TJ, Kulin HE, Zaino RJ. Peutz-Jeghers syndrome. Am J Gastroenterol 2000;95:596-604. [Crossref] [PubMed]
- Algarni M, Raml E, Trabulsi N, et al. Small bowel intussusception and concurrent sigmoid polyp with malignant transformation in Peutz-Jeghers syndrome. J Surg Case Rep 2019;2019:rjz004. [Crossref] [PubMed]
- Peutz JLA. On a very remarkable case of familial polyposis of mucous membrane of intestinal tract and nasopharynx accompanied by peculiar pigmentations of skin and mucous membrane. Ned Tijdschr Geneeskd 1921;10:134-46.
- Jeghers H, McKusick VA, Katz KH. Generalized intestinal polyposis and melanin spots of the oral mucosa, lips and digits; a syndrome of diagnostic significance. N Engl J Med 1949;241:993-illust. passim. [Crossref] [PubMed]
- Daniell J, Plazzer JP, Perera A, et al. An exploration of genotype-phenotype link between Peutz-Jeghers syndrome and STK11: a review. Fam Cancer 2018;17:421-7. [Crossref] [PubMed]
- Sweet K, Willis J, Zhou XP, et al. Molecular classification of patients with unexplained hamartomatous and hyperplastic polyposis. JAMA 2005;294:2465-73. [Crossref] [PubMed]
- Borrego S, Wright FA, Fernández RM, et al. A founding locus within the RET proto-oncogene may account for a large proportion of apparently sporadic Hirschsprung disease and a subset of cases of sporadic medullary thyroid carcinoma. Am J Hum Genet 2003;72:88-100. [Crossref] [PubMed]


