
The expression of SOX9, Tiam1, and PTEN is correlated with angiogenesis and prognosis in gastric cancer
Introduction
Improved living standards and increased social pressure have seen a subsequent year-by-year rise in the incidence of gastric cancer (GC) and a drop in the age of patients with the disease. Even as a common malignant tumor, GC is often missed because of its atypical early-stage symptoms, which make it difficult to distinguish from other gastric diseases, such as gastric ulcers. As a consequence, many patients are already at an advanced-stage of GC by the time they receive treatment (1). With high rates of metastasis and recurrence and a poor prognosis, discovering effective molecular markers for predicting the prognosis of GC patients is crucial. Studies have reported abnormal expressions of SOX9, Tiam1, and PTEN are present in GC tissues (2-5), which may bear significance for early diagnosis and predicting prognosis in GC. Neovascularization is an essential process in tumor growth, invasion, and metastasis. However, there are few literatures to comprehensively analyze the expression of SOX9, Tiam1 and PTEN in gastric cancer and their clinical significance. In the progression of the disease, angiogenesis plays an important role in tumor growth and tumor invasion and metastasis. VEGF is the best known angiogenic factor, which plays an important role in tumor angiogenesis (6). Based on this, we detected the protein expression of SOX9, Tiam1, PTEN, and VEGF in GC tissues by immunohistochemistry and explored the correlation between the expression of SOX9, Tiam1, and PTEN and GC angiogenesis and prognosis. We present the following article in accordance with the REMARK reporting checklist (available at http://dx.doi.org/10.21037/tcr-20-2071).
Methods
Study subjects
Patients with GC who underwent surgery in our hospital between January, 2017 and October, 2018 were enrolled in the study. The GC diagnosis of each patient was confirmed by pathology. A total of 90 patients (52 males and 38 females) with an average age of (56.69±8.32) years old (range: 32 to 78 years) were included. In terms of TNM staging, there were 12 cases of stage I, 24 cases of stage II, 35 cases of stage III, and 19 cases of stage IV. Lymph node metastasis was present in 35 cases. In 34 cases, the tumors had moderate to high differentiation, while 56 cases had a low degree of differentiation. Normal tissues from the 90 patients (taken more than 3 cm from the edge of the cancer) were collected as the control group. All patients had complete clinical and case data, and none of the patients received radiotherapy or chemotherapy before surgery. The trial was conducted in accordance with the Declaration of Helsinki. This study was approved by the Ethics Committee of our hospital and the informed consent was obtained from the patients.
Research methods
The surgically resected GC specimens were fixed with 10% formaldehyde solution, embedded in paraffin, and sliced at 4 µm. The expression levels of SOX9, Tiam1, PTEN, and VEGF were detected by immunohistochemistry. SOX9, Tiam1, and PTEN proteins were mainly localized in the cytoplasm and appeared as brownish-yellow particles. The positive expression was judged based partly on comprehensive evaluation of the depth of color: no color (0 points); light yellow (1 point); and brownish yellow (2 points). It was also judged as positive based on the percentage of positive cells: ≤10% (1 point); between 11% and 50% (2 points); between 51% and 75% (3 points); and ≥75% (4 points). The two scores were multiplied to analyze the result, according to the following thresholds: >3 as positive, and ≤3 as negative.
After surgery, the patients were followed-up every 3 months in the first year after the operation, and every six months in the two years after that. The patients’ survival was recorded over a follow-up period lasting for 3 years.
Statistical analysis
The count data were expressed as rate (%) and analyzed using the χ2 test in SPSS 22.0 (IBM, USA). The correlations between the expressions of SOX9, Tiam1, PTEN, and VEGF were analyzed using Spearman’s correlation analysis. Kaplan-Meier survival curves were generated for each group, and the survival rates between groups were compared using the log-rank method. A P value of <0.05 was considered to be statistically significant.
Results
Expression of SOX9, Tiam1, PTEN, and VEGF in GC and normal tissues
The positive expression rates of SOX9, Tiam1, PTEN, and VEGF in GC tissues were 75.56%, 61.11%, 52.22%, and 48.89%, respectively, compared with 6.67%, 4.44%, 97.78%, and 2.22% in the normal adjacent tissues, respectively. In the GC tissues, the positive expression rates of SOX9, Tiam1, and VEGF were significantly higher than in the adjacent normal tissues; however, the positive expression rate of PTEN was significantly lower than in the adjacent normal tissues (P<0.05, Figure 1, Table 1).
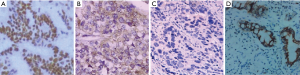
Table 1
| Group | Cases | SOX9 (+) | Tiam1 (+) | PTEN (+) | VEGF (+) |
|---|---|---|---|---|---|
| Gastric cancer tissue | 90 | 68/75.56 | 55/61.11 | 47/52.22 | 44/48.89 |
| Adjacent tissues | 90 | 6/6.67 | 4/4.44 | 88/97.78 | 2/2.22 |
| χ2 | 88.210 | 65.581 | 49.807 | 45.057 | |
| P | 0.000 | 0.000 | 0.000 | 0.000 |
VEGF, vascular endothelial growth factor.
Correlation of SOX9, Tiam1, PTEN, and VEGF in GC
According to the results of Spearman’s correlation analysis, the expression of SOX9 (r=0.349, P=0.001) and Tiam1 (r=0.370, P=0.000) in GC tissue was positively correlated with the expression of VEGF, while the expression of PTEN was negatively correlated with the expression of VEGF (r=−0.311, P=0.000; Table 2).
Table 2
| Index | Cases | VEGF (+) | VEGF (−) | r | P |
|---|---|---|---|---|---|
| SOX9 (+) | 68 | 40 | 28 | 0.349 | 0.001 |
| SOX9 (−) | 22 | 5 | 17 | ||
| Tiam1 (+) | 55 | 35 | 20 | 0.370 | 0.000 |
| Tiam1 (−) | 35 | 9 | 26 | ||
| PTEN (+) | 47 | 16 | 31 | −0.311 | 0.003 |
| PTEN (−) | 43 | 28 | 15 |
VEGF, vascular endothelial growth factor.
The relationship between the expression of SOX9, Tiam1, and PTEN in GC tissues and the clinicopathological factors
There was no significant difference in the expression of SOX9, Tiam,1 and PTEN in GC tissues of patients according to gender or age (P>0.05). Low tumor differentiation, lymph node metastasis, and high TNM stage were correlated with significantly increased positive expression rates of SOX9 and Tiam1 and significantly decreased positive expression of PTEN (P<0.05, Table 3).
Table 3
| Clinical features | Cases | SOX9 (+) | Tiam1 (+) | PTEN (+) | |||||
|---|---|---|---|---|---|---|---|---|---|
| N | χ2/P | N | χ2/P | N | χ2/P | ||||
| Gender | 0.125/0.724 | 0.116/0.733 | 0.004/0.947 | ||||||
| Men | 52 | 40 | 31 | 27 | |||||
| Women | 38 | 28 | 24 | 20 | |||||
| Age (years) | 1.039/0.308 | 1.102/0.294 | 0.019/0.890 | ||||||
| <60 | 53 | 38 | 30 | 28 | |||||
| ≥60 | 37 | 30 | 25 | 19 | |||||
| Differentiated degree | 4.757/0.029 | 5.424/0.020 | 4.284/0.038 | ||||||
| Low differentiation | 34 | 30 | 26 | 13 | |||||
| Moderate-high differentiation | 56 | 38 | 29 | 34 | |||||
| Lymph node metastasis | 5.253/0.022 | 4.192/0.041 | 7.385/0.007 | ||||||
| Yes | 35 | 31 | 26 | 12 | |||||
| No | 55 | 37 | 29 | 35 | |||||
| TNM stage | 9.636/0.002 | 4.870/0.027 | 5.017/0.025 | ||||||
| I–II | 36 | 21 | 17 | 24 | |||||
| III–IV | 54 | 47 | 38 | 23 | |||||
The relationship between the expression of SOX9, Tiam1, and PTEN and GC prognosis
Based on the follow-up of the GC patients in our study, the survival curve showed that the 3-year survival rates of the groups with positive SOX9 and Tiam1 expression (54.41% and 49.09%, respectively) were significantly lower than those of the groups with negative SOX9 and Tiam1 expression (77.27% and 77.13%, respectively). Meanwhile the 3-year survival rate was significantly higher in the PTEN positive expression group (76.60%) than in the PTEN negative expression group (41.80%). The log-rank test was performed and indicated that there was a significant difference in the survival rates of patients with positive and negative indexes of SOX9, Tiam1, and PTEN (P<0.05, Figure 2, Table 4).

Table 4
| Index | Cases | 3-year survival rate (%) | Log-rank χ2 | P |
|---|---|---|---|---|
| SOX9 | 3.888 | 0.048 | ||
| + | 68 | 54.41% | ||
| − | 22 | 77.27% | ||
| Tiam1 | 8.201 | 0.004 | ||
| + | 55 | 49.09% | ||
| − | 35 | 77.13% | ||
| PTEN | 12.720 | 0.000 | ||
| + | 47 | 76.60% | ||
| − | 43 | 41.80% |
Discussion
GC, with its high incidence, causes more deaths than any other type of digestive tract tumor. GC patients encounter very different treatment effects and prognoses. Still under discussion, the pathogenesis of GC is complex, and it involves multiple genes and multi-stage processes (7). SOX9, which is part of the SOXE subfamily, is one of the most widely studied transcription factors in the SOX family. SOX9 is widely expressed in tissues, including the adult heart and brain. Some researchers have pointed out that it may be involved in the occurrence and development of various cancers, such as intestinal cancer (8). Tiam1 is a ubiquitous guanine nucleotide conversion factor, which is mainly expressed in normal tissues such as human brain and testis; however, its expression in other tissues is low or non-existent. Previous studies have shown that Tiam1 is abundantly expressed in various tumor cell types and is closely related to tumor invasion and metastasis (9). Meanwhile, PTEN is currently the first tumor suppressor gene with phosphatase activity and can negatively regulate the PI3K/AKT signaling pathway. Studies have revealed that a loss of heterozygosity of PTEN occurs in different malignant tumor cells, which may play an important role in tumor occurrence and development (10). In line with previous studies, the results of this study showed that compared with adjacent normal tissues, the positive expression rates of SOX9 and Tiam1 in GC tissues were significantly increased, while the positive expression rates of PTEN were significantly decreased. These results confirm that SOX9, Tiam1, and PTEN are closely related to the occurrence and development of GC.
As the pivotal positive regulator in the process of angiogenesis, VEGF can promote the division and proliferation of endothelial cells and induce angiogenesis in vivo (11). The results of this study showed that the positive expression rate of VEGF was significantly higher in GC tissues than adjacent normal tissues. More extensive analysis of the correlation between SOX9, Tiam1, PTEN, and VEGF in GC tissues revealed that the expression of SOX9 and Tiam1 was positively correlated with the expression of VEGF, while the expression of PTEN was negatively correlated with the expression of VEGF, which suggests that GC involves active angiogenesis (12). So, we speculated that SOX9, Tiam1, and PTEN may affect tumor angiogenesis via the regulation of VEGF expression. The relationship between the expression of SOX9, Tiam1, PTEN and the clinical pathological factors of GC was studied, which showed that the positive expression rate of the three proteins was closely related to the degree of tumor differentiation, lymph node metastasis, and clinical TNM stage. Patients with a low degree of tumor differentiation, lymph node metastasis, or high TNM stage had a significantly increased positive expression rate of SOX9 and Tiam1, but a significantly decreased positive expression rate of PTEN. In addition, the results showed that the three-year survival rates of the groups with positive SOX9 and Tiam1 expression were significantly lower than those of the corresponding negative expression groups. The three-year survival rate was also significantly higher in the positive PTEN expression group than in the negative PTEN expression group. This result suggests that, to a certain extent, the expression of SOX9, Tiam1, and PTEN reflects the prognosis of GC patients: the high expression of SOX9 and Tiam1 or the low expression of PTEN may indicate increased tumor malignancy and disease progression (13-15). Our findings may be explained by the involvement of SOX9, Tiam1, and PTEN in multiple intracellular activities in the occurrence and development of GC. Previous studies have shown that the SOX9 expression may promote the expression of carcinoembryonic antigen-related cell adhesion molecule 1 (CEACAM1). The expression of CEACAM1 in GC tissue can promote the angiogenesis and lymph node metastasis of GC (16,17). Moreover, Tiam1 plays an important role in tumor invasion and metastasis by affecting the cytoskeletal structure, cell adhesion, and other activities via the Tiam1-Rac signaling pathway, while PTEN can inhibit tumor cell migration and metastasis via regulating the PI3K/AKT signaling pathway (18).
In conclusion, SOX9 and Tiam1 are highly expressed in GC tissues, while the expression of PTEN is low. These three proteins are linearly related to VEGF, which shows that they have a certain effect on angiogenesis in GC. In addition, the protein expression levels of SOX9, Tiam1, and PTEN are closely related to tumor differentiation, lymph node metastasis, clinical TNM staging, and the three-year survival rate of patients with GC. They have important effects on angiogenesis and are closely related to the prognosis of patients with GC, which may provide a certain reference significance for the prediction of prognosis in GC.
Acknowledgments
Funding: None.
Footnote
Reporting Checklist: The authors have completed the REMARK reporting checklist. Available at http://dx.doi.org/10.21037/tcr-20-2071
Data Sharing Statement: Available at http://dx.doi.org/10.21037/tcr-20-2071
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/tcr-20-2071). The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. The trial was conducted in accordance with the Declaration of Helsinki (as revised in 2013). This study was approved by the Ethics Committee of our hospital and the informed consent was obtained from all patients.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- The clinical effect comparison between apatinib and S-1 as the second-line treatment for advanced gastric cancer. Oncol Progress 2017;15:1436-8.
- Liu Z, Yan Y, Cao S, et al. Long non-coding RNA SNHG14 contributes to gastric cancer development through targeting miR-145/SOX9 axis. J Cell Biochem 2018;119:6905-13. [Crossref] [PubMed]
- Chen J, Xu ZY. The expression and correlation of SOX9 and CDX2 genes in the process of gastric carcinogenesis. Int J Dis 2018;38:62-5. (in Chinese.
- Wu SQ, He HQ, Kang Y, et al. MicroRNA-200c affects bladder cancer angiogenesis by regulating the Akt2/mTOR/HIF-1 axis. Transl Cancer Res 2019;8:2713-24. [Crossref]
- Li Z, Yu X, Wang Y, et al. By downregulating TIAM1 expression, microRNA-329 suppresses gastric cancer invasion and growth. Oncotarget 2015;6:17559-69. [Crossref] [PubMed]
- Alexander M, Halmos B. VEGF inhibitors in EGFR-mutated lung cancer: a never-ending story? Ann Transl Med 2018;6:446. [Crossref] [PubMed]
- Katoh H, Ishikawa S. Genomic pathobiology of gastric carcinoma. Pathol Int 2017;67:63-71. [Crossref] [PubMed]
- Cui N, Zhang J, Huang H, et al. Epigenetic regulator ARID1A and stem cell transcription factor SOX9 in the maintenance of pancreatic ductal cell differentiation state and development of intraductal papillary mucinous neoplasia (IPMN) and pancreatic ductal adenocarcinoma (PDAC). Transl Cancer Res 2018;7:S748-51. [Crossref]
- Peng WB, Li XZ. Expression of Tiam1 and Rac1 proteins in invasion and metastasis of gastric carcinoma and its influencing factors. J Clin Exp Med 2018;17:937-40.
- Guan W, Jing Y, Yu L. Prognostic Value of Recurrent Molecular Genetics and Epigenetics Abnormity in T Lymphoblastic Lymphoma/Leukemia—Review. Zhongguo Shi Yan Xue Ye Xue Za Zhi 2017;25:587-91. [PubMed]
- Gong X, Deng ZH, Li CT, et al. Mechanism of action of hypoxia-inducible factor-1, neuroglobin, and vascular endothelial growth factor in promoting cerebral angiogenesis after cerebral contusion and laceration. J Int Neurol Neurosurg 2017;44:10-4.
- Ma JJ, Zou JP, Ma XY. Expression of COX-2, NF-κB and VEGF in gastric carcinoma tissues and its relationship with angiogenesis. Modern J Integrated Chinese Traditional and Western Medicine 2018;27:1263-6.
- Liu T, Zhang L, Zhang F. Expression of SOX7, SOX9 in prostate cancer and its clinical value in diagnosis and prognosis prediction. Acad J PLA Med Sch 2018;39:30-4.
- Peng WB, Tan XQ. Expression and its clinical significance of Tiam 1 and Rac 1 after gastric cancer resection. Clin J Med Offic 2018;46:202-4.
- Zhou XG, Wang K, Shen XG, et al. Effect of miR-214 on PTEN Protein and Its Biological Behavior in Gastric Cancer Cells. Med &Pham J Chin PLA 2018;30:26-30.
- Ashkenazi S, Ortenberg R, Besser M, et al. SOX9 indirectly regulates CEACAM1 expression and immune resistance in melanoma cells. Oncotarget 2016;7:30166-77. [Crossref] [PubMed]
- Qiu YL. Research progress on TIAM1 in pediatric neuroblastoma. Chin J Clin Oncol 2017;44:1160-4.
- Zhang Y, Xiong JB, Chen HP, et al. Progress of PTEN/PI3K/AKT signaling pathway in gastric cancer. Chongqing Medical Journal 2018;47:2601-3.


