
Efficacy of salvage radiotherapy for relapsed/refractory diffuse large B-cell lymphoma
Introduction
Diffuse large B-cell lymphoma (DLBCL) is the most common histological subtype of non-Hodgkin lymphoma (NHL), accounting for approximately 30% of all NHL patients (1,2). Although substantial progress has been made in DLBCL treatment, 50% of DLBCL patients will become refractory or will relapse after treatment (3). High-dose chemotherapy combined with autologous stem cell transplantation (ASCT) has become the standard treatment for patients with relapsed/refractory (R/R) DLBCL (4). However, the prognosis of patients with R/R DLBCL is still unsatisfactory. The median survival time is only 6.3 months, and only 20% of patients survive at 2 years (5).
Radiotherapy (RT) is also one of the main treatment strategies for DLBCL, with a significant standing in the treatment of localized DLBCL (6). However, the role of RT in the salvage treatment of DLBCL is not clear. By retrospectively analyzing the efficacy of RT in R/R DLBCL, this study investigated the standing and role of RT in the salvage treatment of DLBCL and provides significant clinical evidence.
Methods
Diagnostic criteria for patients with R/R DLBCL
Relapsed lymphoma was defined as DLBCL in the presence of any new lesion after the initial complete remission (CR). Refractory lymphoma was defined as meeting any of the following: (I) tumor shrinkage <50% or disease progression (DP) after completion of four courses of standardized chemotherapy; (II) CR achieved after standard chemotherapy but tumor relapse within 6 months; (III) tumor relapse at least twice after CR or (IV) relapse after hematopoietic stem cell transplantation.
Patient characteristics
We retrospectively analyzed 32 patients with R/R DLBCL undergoing salvage RT between January 2009 and June 2016 in our hospital. This study was approved by the Ruijin Hospital Ethics Committee of the Shanghai Jiao Tong University School of Medicine. The need for individual consent was waived by the committee due to the retrospective nature of the study; and data are entered directly onto a database which will not contain personal information. The median follow-up duration of all patients was 25.4 months, with a range of 0.4–98.9 months. We acquired patient treatment, relapse and survival status through follow-up data and analyzed different factors including age, International Prognostic Index (IPI) score, tumor location, staging, chemotherapy regimen prior to RT, RT dose and adverse reactions. Among the 32 patients, 16 were male (50.0%) and 16 were female (50.0%). The median age was 47 years (range, 13–85 years). Pathology confirmed that the primary tumor in all patients was DLBCL. Immunohistochemistry showed CD20-positive reactivity. Overall, 28 patients had refractory DLBCL, and four patients had relapsed DLBCL. At the initial diagnosis, 1 patient was at stage I (3.1%), 12 patients were at stage II (37.5%), 9 patients were at stage III (28.1%), and 10 patients were at stage IV (31.3%). The median IPI score of all patients was 3 points (range, 0–4). Twenty patients (62.5%) had large tumors. At the time of initial diagnosis, all patients received first-line chemotherapy based on CHOP (cyclophosphamide + adriamycin + vincristine + prednisone), and 27 patients also received first-line rituximab targeted therapy. The median number of courses of chemotherapy was 6 (range, 2–16). Chemotherapy regimens mainly included CHOP, ICE (ifosfamide + carboplatin + VP16) and DHAP (cisplatin + cytarabine + dexamethasone). The clinicopathological characteristics of the 32 patients are shown in Table 1.
Table 1
| Clinicopathological data | Number of cases (n=32) |
|---|---|
| Gender | |
| Male | 16 |
| Female | 16 |
| Median age (years) | 47 [13–85] |
| <60 | 22 |
| ≥60 | 10 |
| Staging at the time of diagnosis | |
| I | 1 |
| II | 12 |
| III | 9 |
| IV | 10 |
| Relapsed/refractory | |
| Refractory | 28 |
| Relapsed | 4 |
| Serum LDH | |
| Normal | 7 |
| Elevated | 25 |
| B symptoms | |
| No | 23 |
| Yes | 9 |
| Tumor diameter | |
| ≤5 cm | 7 |
| >5 cm | 20 |
| Unclear | 5 |
| Extranodal invasion | |
| No | 5 |
| Yes | 27 |
| Lesion location | |
| Head and neck | 7 |
| Thorax and mediastinum | 10 |
| Axilla | 2 |
| Digestive tract | 2 |
| Abdominal and pelvic lymph nodes | 7 |
| Skeleton | 5 |
| Soft tissue | 2 |
| Other | 1 |
| International Prognostic Index (IPI) score | |
| 0 | 1 |
| 1 | 6 |
| 2 | 8 |
| 3 | 14 |
| 4 | 3 |
| Number of previous chemotherapy regimens | |
| 1 | 22 |
| 2 | 7 |
| 3 | 2 |
| 4 | 1 |
| First-line treatment | |
| CHOP | 5 |
| R-CHOP | 27 |
| Response to first-line chemotherapy | |
| CR | 1 |
| PR | 8 |
| SD | 18 |
| DP | 5 |
| Number of chemotherapy treatments [range] | 6 [2–16] |
DLBCL, diffuse large B-cell lymphoma; LDH, lactate dehydrogenase; CR, complete remission; PR, partial remission; SD, stable disease; DP, disease progression .
Statistical analysis
SPSS 19.0 software was used for statistical analysis. The Kaplan-Meier test and the log-rank test were used for univariate analyses. The Cox regression test was used for multivariate analysis to determine the association between clinicopathological characteristics and relapse type. P<0.05 was considered statistically significant.
Results
RT type
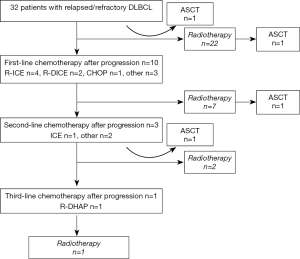
The median duration from the first treatment to RT was 9.3 months (range, 4.0–77.9 months). The median radiation dosage was 42.7 Gy (range, 30–54 Gy). Overall, 4 patients (12.5%) underwent two-dimensional RT, 2 patients (6.3%) underwent three-dimensional conformal RT (3D/CRT), and 26 patients (81.3%) underwent intensity-modulated RT (IMRT). One patient (3.1%) received expanded field RT, 14 (43.8%) received involved field RT (IFRT), and 17 (53.1%) received involved site RT (ISRT). Among all patients, 28 patients had one lesion at the time of RT and four patients had two isolated lesions. All patients with residual lesions underwent RT.
Four patients (12.5%) underwent ASCT, among whom two underwent stem cell transplantation prior to RT and two underwent stem cell transplantation after RT. The treatment flowchart for patients with R/R DLBCL is shown in Figure 1.
Survival analysis
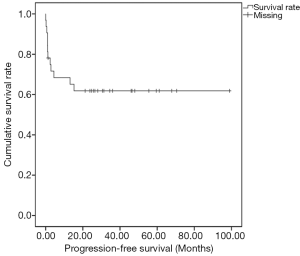
The median follow-up duration of all patients was 25.4 months (range, 0.4–98.9 months). After RT, 13 patients (40.6%) had achieved CR or partial remission (PR), 10 patients (31.3%) had stable disease (SD), and 9 patients (28.1%) had DP after RT. In the end, five patients died due to DP. The 5-year progression-free survival (PFS) and overall survival (OS) of the 32 patients were 61.8% (Figure 2) and 83.2%, respectively.
The relationship between clinicopathological factors and survival
Univariate analysis showed that the patients’ IPI score and the presence of a single lesion at the time of RT were associated with 5-year PFS (P=0.007 and P<0.001, respectively) (Table 2). Multivariate analysis showed that multiple lesions were an independent factor for poor prognosis in patients with R/R DLBCL (P=0.005) (Table 2).
Table 2
| Clinicopathological factors | Cases | Univariate analysis | Multivariate analysis | |||
|---|---|---|---|---|---|---|
| 5-year PFS (%) | P value | HR value (95% CI) | P value | |||
| Gender | 0.618 | |||||
| Male | 16 | 68.2 | ||||
| Female | 16 | 56.3 | ||||
| Age (years) | 0.809 | |||||
| <60 | 22 | 62.6 | ||||
| ≥60 | 10 | 60.0 | ||||
| Staging at the time of diagnosis | 0.178 | |||||
| I–II | 13 | 75.5 | ||||
| III–IV | 19 | 52.6 | ||||
| Serum LDH | 0.183 | |||||
| Normal | 7 | 85.7 | ||||
| Elevated | 25 | 54.9 | ||||
| B symptoms | 0.750 | |||||
| No | 23 | 64.5 | ||||
| Yes | 9 | 55.6 | ||||
| Tumor diameter | 0.526 | |||||
| ≤5 cm | 7 | 71.4 | ||||
| >5 cm | 20 | 58.9 | ||||
| Extranodal invasion | 0.429 | |||||
| No | 5 | 75.0 | ||||
| Yes | 27 | 59.3 | ||||
| IPI index | 0.007 | 0.77 | ||||
| 3–4 | 17 | 41.2 | 4.249 (0.857–21.081) | |||
| 0–2 | 15 | 85.7 | 1 | |||
| Relapsed/refractory | 0.095 | |||||
| Refractory | 28 | 67.2 | ||||
| Relapse | 4 | 25.0 | ||||
| Previous chemotherapy regimen number | 0.600 | |||||
| 1 | 22 | 63.6 | ||||
| 2–4 | 10 | 58.3 | ||||
| Response to first chemotherapy | 0.176 | |||||
| CR and PR | 9 | 44.4 | ||||
| SD and DP | 23 | 68.6 | ||||
| Single lesion | <0.001 | 0.005 | ||||
| No | 4 | 0.0 | 23.998 (2.580–223.235) | |||
| Yes | 28 | 70.7 | 1 | |||
| Radiotherapy dose (Gy) | 0.268 | |||||
| <45 | 17 | 70.6 | ||||
| ≥45 | 15 | 51.3 | ||||
| Radiotherapy exposure field | 0.194 | |||||
| IFRT or expanded field | 15 | 73.3 | ||||
| ISRT | 17 | 51.3 | ||||
| Radiotherapy technology | 0.867 | |||||
| 2D or 3D/CRT | 6 | 66.7 | ||||
| IMRT | 26 | 60.7 | ||||
| Stem cell transplantation | 0.604 | |||||
| No | 28 | 63.6 | ||||
| Yes | 4 | 50.0 | ||||
DLBCL, diffuse large B-cell lymphoma; LDH, lactate dehydrogenase; PR, partial remission; SD, stable disease; DP, disease progression; IPI, International Prognostic Index; IFRT, involved field radiotherapy; ISRT, involved site radiotherapy.
Toxic reaction
Toxic reactions in most patients at the early stage of RT were mild. Seven patients (21.9%) were at Common Terminology Criteria for Adverse Events (CTCAE) level 1, and seven patients (21.9%) were at CTCAE level 2. Adverse reactions mainly included neutropenia, diarrhea, dermatitis, mucositis and dysphagia. One patient had grade 3 neutropenia. There were no significantly long-term toxic reactions.
Discussion
Although substantial progress has been made for the treatment of DLBCL, the majority of patients cannot be cured through regular treatment. High-dose chemotherapy combined with ASCT can improve survival and is currently the standard treatment for patients with R/R DLBCL (4). However, less than 10% of patients achieve long-term disease-free survival after second-line treatment (7). For patients with R/R DLBCL who are not suitable for direct ASCT second-line salvage treatment is still the primary option. Therefore, exploration of an economical salvage treatment plan with high efficiency and low toxicity has extensive clinical applications (8).
High-dose chemotherapy combined with ASCT can improve CR and OS rates (9). In the current study, four patients underwent ASCT: two prior to RT and two after RT. Two of these four patients (50%) had CR. However, currently, prospective evidence is still lacking for RT indications of patients with R/R DLBCL undergoing ASCT and the sequence for RT and ASCT (10,11). In most studies, patients with large tumors or with large lesions after chemotherapy undergo RT, including preventive RT (11,12). Therefore, the indications, dosage and reactions of RT are not clear. However, we found from previous studies that RT both prior to and after ASCT are beneficial for local control and may also extend PFS and OS (10,11,13,14). The main advantage of RT is the improvement of local control, which significantly reduces the relapse rate prior to and after transplantation (10,12). This advantage still exists in the era of anti-CD20 monoclonal antibodies. Although currently relevant studies on dose comparison are lacking, the effective dose of RT is approximately 30–40 Gy. The data in our study also showed that RT doses higher than 45 Gy did not further extend survival. Additionally, IFRT did not increase RT-related toxicity.
Another noteworthy issue is that in this study, multiple lesions at the time of RT were the only independent prognostic factor affecting PFS (P=0.005). This result indirectly indicates that the reactions of patients with R/R DLBCL to RT were associated with staging at the time of RT. Patients with more advanced staging at the time of RT had poorer prognoses. The number of patients with multiple lesions at the time of RT was limited (12.5%) in this study, and thus, more patients are needed to confirm our finding.
IPI has always been a recognized prognostic criterion for DLBCL, including age, ECOG score, Ann Arbor staging, lactate dehydrogenase (LDH) levels and degree of extranodal involvement. IPI is still a powerful prognostic indicator in the anti-CD20 monoclonal antibody era (15). Univariate analysis of the patients in this study showed that moderate to high risk in the IPI score was a negative factor for PFS. However, in the following multivariate analysis, we did not identify any correlation between IPI and PFS. An unpublished trial, DSHNHL 2004-3, assessed the effects of consolidative RT in patients aged 18–60 years with IPI scores of 1 or 0 and large tumors (>7.5 cm). This trial assessed consolidative RT after chemotherapy and found that the prognosis of patients without RT was significantly poorer than patients undergoing RT, indicating that RT may be beneficial for patients with low IPI scores. Due to the limited sample size in this study, our finding should be confirmed by studies with larger sample sizes.
A single-center study retrospectively analyzed 110 patients who underwent RT due to R/R invasive NHL and assessed the reaction rate and time to local relapse. In that study, 77% of the patients had DLBCL (16). Approximately 85% of tumors entered remission after a median radiation dose of 37.8 Gy, and 66% of tumors were under local control at 5 years. In our study, the 5-year PFS and OS of the 32 patients were 61.8% and 83.2%, respectively, which is similar to the previous study. Thus, we conclude that RT may improve the prognosis of patients with R/R DLBCL.
Although this is a retrospective study with limited sample size, our analysis showed that salvage therapy may extend survival with mild adverse reactions for strictly selected patients with R/R DLBCL, such as patients with a single isolated residual or relapsed lesion. Prospective studies with larger sample sizes are needed to confirm our finding.
Acknowledgments
Funding: Supported in part by
Footnote
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/tcr.2019.06.12). The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. The study was conducted in accordance with the Declaration of Helsinki (as revised in 2013). This study was approved by the Ruijin Hospital Ethics Committee of the Shanghai Jiao Tong University School of Medicine. The need for individual consent was waived by the committee due to the retrospective nature of the study.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Morton LM, Wang SS, Devesa SS, et al. Lymphoma incidence patterns by WHO subtype in the United States, 1992-2001. Blood 2006;107:265-76. [Crossref] [PubMed]
- van Leeuwen MT, Turner JJ, Joske DJ, et al. Lymphoid neoplasm incidence by WHO subtype in Australia 1982-2006. Int J Cancer 2014;135:2146-56. [Crossref] [PubMed]
- Sehn LH, Berry B, Chhanabhai M, et al. The revised International Prognostic Index (R-IPI) is a better predictor of outcome than the standard IPI for patients with diffuse large B-cell lymphoma treated with R-CHOP. Blood 2007;109:1857-61. [Crossref] [PubMed]
- Oliansky DM, Czuczman M, Fisher RI, et al. The role of cytotoxic therapy with hematopoietic stem cell transplantation in the treatment of diffuse large B cell lymphoma: update of the 2001 evidence-based review. Biol Blood Marrow Transplant 2011;17:20-47.e30. [Crossref] [PubMed]
- Crump M, Neelapu SS, Farooq U, et al. Outcomes in refractory diffuse large B-cell lymphoma: results from the international SCHOLAR-1 study. Blood 2017;130:1800-8. [Crossref] [PubMed]
- Miller TP, Dahlberg S, Cassady JR, et al. Chemotherapy alone compared with chemotherapy plus radiotherapy for localized intermediate- and high-grade non-Hodgkin's lymphoma. N Engl J Med 1998;339:21-6. [Crossref] [PubMed]
- Gisselbrecht C, Glass B, Mounier N, et al. Salvage regimens with autologous transplantation for relapsed large B-cell lymphoma in the rituximab era. J Clin Oncol 2010;28:4184-90. [Crossref] [PubMed]
- Thieblemont C, Gisselbrecht C. Second-line treatment paradigms for diffuse large B-cell lymphomas. Curr Oncol Rep 2009;11:386-93. [Crossref] [PubMed]
- Philip T, Guglielmi C, Hagenbeek A, et al. Autologous bone marrow transplantation as compared with salvage chemotherapy in relapses of chemotherapy-sensitive non-Hodgkin's lymphoma. N Engl J Med 1995;333:1540-5. [Crossref] [PubMed]
- Hoppe BS, Moskowitz CH, Filippa DA, et al. Involved-field radiotherapy before high-dose therapy and autologous stem-cell rescue in diffuse large-cell lymphoma: long-term disease control and toxicity. J Clin Oncol 2008;26:1858-64. [Crossref] [PubMed]
- Biswas T, Dhakal S, Chen R, et al. Involved field radiation after autologous stem cell transplant for diffuse large B-cell lymphoma in the rituximab era. Int J Radiat Oncol Biol Phys 2010;77:79-85. [Crossref] [PubMed]
- Hoppe BS, Moskowitz CH, Zhang Z, et al. The role of FDG-PET imaging and involved field radiotherapy in relapsed or refractory diffuse large B-cell lymphoma. Bone Marrow Transplant 2009;43:941-8. [Crossref] [PubMed]
- Friedberg JW, Neuberg D, Monson E, et al. The impact of external beam radiation therapy prior to autologous bone marrow transplantation in patients with non-Hodgkin's lymphoma. Biol Blood Marrow Transplant 2001;7:446-53. [Crossref] [PubMed]
- Wendland MM, Smith DC, Boucher KM, et al. The impact of involved field radiation therapy in the treatment of relapsed or refractory non-Hodgkin lymphoma with high-dose chemotherapy followed by hematopoietic progenitor cell transplant. Am J Clin Oncol 2007;30:156-62. [Crossref] [PubMed]
- Ziepert M, Hasenclever D, Kuhnt E, et al. Standard International prognostic index remains a valid predictor of outcome for patients with aggressive CD20+ B-cell lymphoma in the rituximab era. J Clin Oncol 2010;28:2373-80. [Crossref] [PubMed]
- Tseng YD, Chen YH, Catalano PJ, et al. Rates and durability of response to salvage radiation therapy among patients with refractory or relapsed aggressive non-Hodgkin lymphoma. Int J Radiat Oncol Biol Phys 2015;91:223-31. [Crossref] [PubMed]


