
Mass size is a major predictor of hypertensive attack during surgery in patients with paraganglioma of retroperitoneum
Introduction
Paraganglioma, known as extra-adrenal pheochromocytoma, can be located retroperitoneum, thorax, head & neck and urinary bladder (UB), etc. (1). It is a notably rare tumor and comprises 18% of pheochromocytomas, with 10% in the bladder (2,3). Paraganglioma of the UB comprises approximately 0.06% of bladder tumors (4).
Paragangliomas also produce catecholamines such as epinephrine, norepinephrine, metanephrine and dopamine. Thus, they can induce severe hypertension, tachycardia and even death. Classical triad clinical symptoms are palpitation, headache, diaphoresis, however, anxiety, panic attack, syncope, abdominal pain and weight loss can appear in some cases (5,6). In addition, paraganglioma of the UB also induces hematuria and urinary attack which are due to catecholamine release caused by micturition or distension of the bladder (7). These symptoms can help diagnosis of paraganglioma, but most patients do not have symptoms and tumor is located outside the adrenal gland, thus, other diagnoses take precedence over paraganglioma and preoperative diagnosis is difficult. Now, the incidental discovery of paraganglioma is becoming common with the widespread use of computed tomography (CT) (8,9).
The only curative treatment of paraganglioma is surgical resection (10). However, surgical manipulation of paraganglioma can induce a massive release of catecholamines leading to fluctuation of blood pressure, arrythmia, myocardial infarction and stroke as like pheochromocytoma (9). Thus, preoperative medication of combined α- and β-adrenergic blocker therapy is necessary to control and prevent intraoperative hypertension. Especially, predicting which patients may have intraoperative instability of blood pressure or hypertension would be useful because it may facilitate selection of a drug regimen wherein patients predicted more likely to have hemodynamic instability during surgery (11,12). Previous studies have found a level of catecholamine as a predictor of hemodynamic instability in pheochromocytoma (13,14). But unlike pheochromocytoma, it is difficult to diagnose paraganglioma before surgery as already mentioned, so it is not easy to test for catecholamine and pretreat medication before surgery. Therefore, it is needed that other factors that can predict hypertensive attack or hemodynamic instability before surgery.
To date, unfortunately, the risk factors for hypertensive attack during surgery are unknown because of tumor’s paucity. To our knowledge, this study is the first study to the evaluate risk factors for hypertensive attack during surgery in patients with retroperitoneal paragangliomas. We present the following article in accordance with the STARD reporting checklist (available at https://tcr.amegroups.com/article/view/10.21037/tcr-22-707/rc).
Methods
Study design and patients
We conducted a retrospective chart review of all 32 patients who had a pathological diagnosis of extra-adrenal paraganglioma between March 2006 and May 2021 in our center. We analyzed the risk factors impacting hypertensive attack in 24 patients with retroperitoneal paragangliomas including 5 UB paragangliomas. The mean patients’ age was 63.8±11.4 years. The male:female ratio was 13:11. The mean systolic and diastolic blood pressure were 128.1±15.4 and 76.8±8.6 mmHg, respectively. The follow-up period was 55.2±48.2 months. The risk factors such as age, sex, height, weight, blood pressure before surgery (systolic and diastolic), history of hypertension, pre-operative symptoms, mass location (bladder vs. retroperitoneum) and mass size were compared between groups with (n=11) and without (n=13) hypertensive attack during surgery.
For the UB paragangliomas, transurethral resection of bladder (TURB) using bipolar current was performed for in three patients, robot assisted partial cystectomy in one patient and open partial cystectomy was performed in one patient with end stage renal disease. For the retroperitoneal mass, laparoscopic and open resection were performed in five and 14 patients, respectively. Complications included massive bleeding (requirement for transfusion of blood) or hypertensive attack during surgery. Hypertension was defined as a systolic blood pressure >140 mmHg or a diastolic blood pressure >90 mmHg. Hypertensive attack was assessed using changes of blood pressure from electric medical chart and was defined as systolic blood pressure >180 mmHg during the excision of the mass (15,16). The size of the paraganglioma was obtained in three dimensions of CT scan, and longest length among them was determined as the size of the mass.
Ethical statement
The study was conducted in accordance with the Declaration of Helsinki (as revised in 2013). This study was approved by our institution review board (IRB) of Ulsan University Hospital (No. UUH-2021-12-009) and individual consent for this retrospective analysis was waived.
Statistical analyses
Statistical analyses were performed using the Statistical Package for the Social Sciences version 21 (IBM Corporation, Armonk, NY, USA). The Wilcoxon signed-rank test, Mann-Whitney U test, and chi-square trend test were used to evaluate the significance of differences between the groups. All statistical analyses were two-sided, and P<0.05 was considered statistically significant. The predictive power was assessed by the area under the curve (AUC) of the receiver operating characteristic (ROC) curve.
Results
There were 32 patients with paraganglioma. Regarding tumor locations, there were 19 retroperitoneal, five UB, two middle-ear cavity, two mediastinal, two neck, one spinal cord, and one duodenal paraganglioma (Figure 1). Of these, we analyzed the 24 patients (75.0%) with retroperitoneal and UB paragangliomas. There was hypertensive attack in 11 (45.8%) of the 24 patients. Four patients experienced massive bleeding during the surgery; however, there were no unusual clinical courses after surgery. Seven (29.2%) of the 24 patients had preoperative symptoms such as pain, fluctuation of blood pressure, and palpable mass (Table 1). There was preoperative history of hypertension in 11 patients (45.8%). The patients with preoperative hypertension had routine anti-hypertensive drugs such as calcium channel block or angiotensin-converting enzyme (ACE) inhibitor or alpha blocker, etc., and control of hypertension. There were five patients with systolic blood pressure >140 mmHg or diastolic blood pressure >90 mmHg, however, none of these patients had hypertensive attack or massive bleeding during surgery. Seven (29.2%) among 24 patients had a hormonal test including catecholamine before surgery. There was no recurrence or disease specific mortality in all 24 patients for an average of 55.2 months after follow-up.
Table 1
| Characteristics | Retroperitoneum including urinary bladder (n=24) |
|---|---|
| Sex (male:female) | 13:11 |
| Age, years, mean ± SD | 63.8±11.4 |
| Body mass index, kg/m2, mean ± SD | 24.3±3.8 |
| History of hypertension, n (%) | 11 (45.8) |
| Pre-operative blood pressure | |
| Systolic, mmHg, mean ± SD | 128.1±15.4 |
| Diastolic, mmHg, mean ± SD | 76.8±8.6 |
| Pre-operative symptoms, n (%) | 7 (29.2) |
| Pain | 4 (16.7) |
| Fluctuation of blood pressure | 2 (8.3) |
| Palpable mass | 1 (4.2) |
| Incidentaloma in CT scan | 17 (70.8) |
| Mass size, cm (longest) | |
| Mean ± SD | 4.1±3.2 |
| Range | 0.6–11.5 |
| Events during surgery, n (%) | |
| Massive bleeding | 4* (16.7) |
| Fluctuation of blood pressure | 11 (45.8) |
| Adrenal-related symptoms | 5 (20.8) |
| Recurrence, n | 0 |
| Disease specific death, n | 0 |
| Follow-up period, months, mean ± SD | 55.2±48.2 |
Adrenal-related symptoms: tachycardia (frequency >100 bpm), desaturation (SpO2 <90%), acidosis (pH <7.3). *, four patients simultaneously had elevation of blood pressure and massive bleeding during surgery. SD, standard deviation; CT, computed tomography.
Between patients with (n=11) and without (n=13) hypertensive attack, there was a significant difference in mass size (5.9±3.4 vs. 2.6±2.1 cm, P=0.007). The mass location also significantly differed between the two groups (P=0.018). However, the mean size of the bladder mass was significantly smaller than that of the retroperitoneal mass (1.59±1.2 vs. 4.8±3.2 cm, P=0.002). There was no difference in age, sex, height, weight, blood pressure, hypertension history, or presence of preoperative symptoms between the two groups (Table 2).
Table 2
| Variables | No complicated events (n=13) | Hypertensive attack (n=11) | P value |
|---|---|---|---|
| Age, years, mean ± SD | 62.7±11.3 | 65.0±12.0 | 0.635 |
| Sex (male:female) | 8:5 | 5:6 | 0.454 |
| Height, cm, mean ± SD | 163.0±7.2 | 161.4±9.1 | 0.623 |
| Weight, kg, mean ± SD | 64.3±10.4 | 62.7±11.9 | 0.747 |
| Blood pressure | |||
| Systolic, mmHg, mean ± SD | 129.2±17.0 | 126.8±13.8 | 0.714 |
| Diastolic, mmHg, mean ± SD | 76.2±7.2 | 77.5±10.3 | 0.727 |
| History of hypertension, n (%) | 6 (46.2) | 5 (45.5) | 0.698 |
| Pre-operative symptoms, n (%) | 2 (15.4) | 5 (45.5) | 0.142 |
| Mass location, n (%) | 0.018 | ||
| Bladder | 5 (38.5) | 0 (0) | |
| Retroperitoneum | 8 (61.5) | 11 (100.0) | |
| Mass size, cm, mean ± SD | 2.6±2.1 | 5.9±3.4 | 0.007 |
SD, standard deviation.
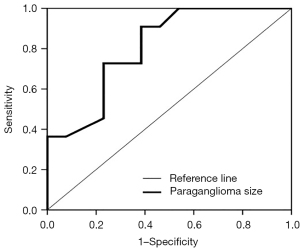
ROC analysis showed that the AUC for predicting surgical complications according to mass size was 0.808 (Figure 2). At cutoff mass size of 4.25 cm, the sensitivity and the specificity were 72.7% and 76.9%, respectively (95% CI: 0.635–0.981).
Discussion
Paraganglioma can present with various features such as pain, hypertension, and palpitation like pheochromocytoma. In this study, most patients had no symptoms, and the paraganglioma was incidentally found with CT or during surgery. Moreover, only seven patients underwent preoperative blood and urine evaluation for catecholamine to diagnose paraganglioma. For patients with pheochromocytoma, an adrenal mass is usually diagnosed imaging studies; thereafter, biochemical testing is performed in all patients with adrenal mass before surgery (17). This difficulty in diagnosing paraganglioma before surgery unlike for pheochromocytoma, has made the un-predicting risk of complications such as hypertensive attack and massive bleeding during surgery (9). Therefore, to prevent these complications, we evaluate preoperative risk factors for hypertensive attack in patients with retroperitoneal paragangliomas, because predicting hypertensive attack is essential.
According to the results in the current study, all patients with symptoms had retroperitoneal paraganglioma. In addition, events during surgery occurred only in cases of retroperitoneal paraganglioma. However, it seems that this difference was not due to the location, but the size of the paraganglioma. Thus, history taking is important, when paraganglioma is suspected, especially if a huge mass (>4.25 cm) is noted. Thereafter endocrinologic evaluation and treatment before surgery are required. Functional imaging such as meta-iodobenzylguanidine (MIBG) scan and positron emission tomography (PET)-CT is also helpful (18,19). Moreover, preoperative combined α- and β-adrenergic blocker therapy can reduce the hypertensive attack during surgery (5,20). Furthermore, in these patients, operators should be careful during surgical manipulation.
There were a few limitations to this study. First, the number of patients was small because paraganglioma is notably rare. Therefore, multicenter studies are required for more accurate results. Second, the current study was retrospective. However, a prospective study of paraganglioma would be complex because the diagnosis of paraganglioma is difficult and incidental. Third, the sizes of UB paraganglioma in the current study were small, as such we could not confirm if the huge UB paraganglioma could induce intraoperative events. However, there are some case reports on this subject. Kurose et al. reported on a 32-mm UB paraganglioma (21). At TURB initiation, the patient’s systolic blood pressure was 120 mmHg but was acutely elevated to over 200 mmHg, and the tumor easily bled. Falcão et al. also reported a UB paraganglioma that underwent by transurethral resection with blood pressure elevation (22).
There are a few similar studies on risk factors for hemostatic instability during pheochromocytoma surgery. In those studies, risk factors aware large tumor size and high urine catecholamine levels (12,13,23). Since pheochromocytoma and paraganglioma are histologically the same tumors, it is expected that they show similar patterns. Thus, urine catecholamines should be considered in further studies.
Pheochromocytoma and paraganglioma are the most heritable tumors and autosomal dominant hereditary cancer diseases (24). In our country, generally genetic screening is not recommended for patients with paraganglioma although clinical practice guidelines of paraganglioma recommend testing for germline variants in all patients (25,26). First, gene sequencing is expensive in our country. Second, there was a just single tumor in retroperitoneum and bladder in all patients. Third, there was no signs such as multiple endocrine neoplasia type 2 caused by RET gene, neurofibromatosis type I caused by NF1 gene, and von Hippel-Lindau disease caused by VHL gene in all patients.
Conclusions
Mass size impacted occurrence of hypertensive attack during surgery in patients with retroperitoneal paraganglioma. Surgeons have to be watchful regarding of intraoperative hypertension during resection of retroperitoneal masses exceeding 4.25 cm, which are suspected as paraganglioma.
Acknowledgments
Funding: None.
Footnote
Reporting Checklist: The authors have completed the STARD reporting checklist. Available at https://tcr.amegroups.com/article/view/10.21037/tcr-22-707/rc
Peer Review File: Available at https://tcr.amegroups.com/article/view/10.21037/tcr-22-707/prf
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at https://tcr.amegroups.com/article/view/10.21037/tcr-22-707/coif). The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. The study was conducted in accordance with the Declaration of Helsinki (as revised in 2013). This study was approved by our institution review board (IRB) of Ulsan University Hospital (No. UUH-2021-12-009) and individual consent for this retrospective analysis was waived.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Leung AA, Pasieka JL, Hyrcza MD, et al. Epidemiology of pheochromocytoma and paraganglioma: population-based cohort study. Eur J Endocrinol 2021;184:19-28. [Crossref] [PubMed]
- Leestma JE, Price EB Jr. Paraganglioma of the urinary bladder. Cancer 1971;28:1063-73. [Crossref] [PubMed]
- Purnell S, Sidana A, Maruf M, et al. Genitourinary paraganglioma: Demographic, pathologic, and clinical characteristics in the surveillance, epidemiology, and end results database (2000-2012). Urol Oncol 2017;35:457.e9-457.e14. [Crossref] [PubMed]
- Katiyar R, Dwivedi S, Trivedi S, et al. Non-Functional Paraganglioma of the Urinary Bladder Treated by Transurethral Resection: Report of Two Cases. J Clin Diagn Res 2016;10:XD01-3. [Crossref] [PubMed]
- Lenders JW, Eisenhofer G, Mannelli M, et al. Phaeochromocytoma. Lancet 2005;366:665-75. [Crossref] [PubMed]
- Mishra A, Mehrotra PK, Agarwal G, et al. Pediatric and adolescent pheochromocytoma: clinical presentation and outcome of surgery. Indian Pediatr 2014;51:299-302. [Crossref] [PubMed]
- Zhai H, Ma X, Nie W, et al. Paraganglioma of the Urinary Bladder: A Series of 22 Cases in a Single Center. Clin Genitourin Cancer 2017;15:e765-71. [Crossref] [PubMed]
- Gruber LM, Hartman RP, Thompson GB, et al. Pheochromocytoma Characteristics and Behavior Differ Depending on Method of Discovery. J Clin Endocrinol Metab 2019;104:1386-93. [Crossref] [PubMed]
- Neumann HPH, Young WF Jr, Eng C. Pheochromocytoma and Paraganglioma. N Engl J Med 2019;381:552-65. [Crossref] [PubMed]
- Kalra Y, Agarwal HS, Smith AH. Perioperative management of pheochromocytoma and catecholamine-induced dilated cardiomyopathy in a pediatric patient. Pediatr Cardiol 2013;34:2013-6. [Crossref] [PubMed]
- Jain A, Baracco R, Kapur G. Pheochromocytoma and paraganglioma-an update on diagnosis, evaluation, and management. Pediatr Nephrol 2020;35:581-94. [Crossref] [PubMed]
- Kim JH, Lee HC, Kim SJ, et al. Characteristics of Intraoperative Hemodynamic Instability in Postoperatively Diagnosed Pheochromocytoma and Sympathetic Paraganglioma Patients. Front Endocrinol (Lausanne) 2022;13:816833. [Crossref] [PubMed]
- Ma L, Shen L, Zhang X, et al. Predictors of hemodynamic instability in patients with pheochromocytoma and paraganglioma. J Surg Oncol 2020; Epub ahead of print. [Crossref] [PubMed]
- Kim JH, Lee HC, Kim SJ, et al. Perioperative hemodynamic instability in pheochromocytoma and sympathetic paraganglioma patients. Sci Rep 2021;11:18574. [Crossref] [PubMed]
- Kwon SY, Lee KS, Lee JN, et al. Risk factors for hypertensive attack during pheochromocytoma resection. Investig Clin Urol 2016;57:184-90. [Crossref] [PubMed]
- Urabe F, Kimura S, Iwatani K, et al. Risk Factors for Perioperative Hemodynamic Instability in Pheochromocytoma: A Systematic Review and Meta-Analysis. J Clin Med 2021;10:4531. [Crossref] [PubMed]
- Sawka AM, Jaeschke R, Singh RJ, et al. A comparison of biochemical tests for pheochromocytoma: measurement of fractionated plasma metanephrines compared with the combination of 24-hour urinary metanephrines and catecholamines. J Clin Endocrinol Metab 2003;88:553-8. [Crossref] [PubMed]
- van Berkel A, Pacak K, Lenders JW. Should every patient diagnosed with a phaeochromocytoma have a 123 I-MIBG scintigraphy? Clin Endocrinol (Oxf) 2014;81:329-33. [Crossref] [PubMed]
- Han S, Suh CH, Woo S, et al. Performance of 68Ga-DOTA-Conjugated Somatostatin Receptor-Targeting Peptide PET in Detection of Pheochromocytoma and Paraganglioma: A Systematic Review and Metaanalysis. J Nucl Med 2019;60:369-76. [Crossref] [PubMed]
- Weingarten TN, Cata JP, O'Hara JF, et al. Comparison of two preoperative medical management strategies for laparoscopic resection of pheochromocytoma. Urology 2010;76:508.e6-11. [Crossref] [PubMed]
- Kurose H, Ueda K, Uegaki M, et al. Paraganglioma of the urinary bladder: Case report and literature review. IJU Case Rep 2020;3:192-5. [Crossref] [PubMed]
- Falcão G, Carneiro C, Pinheiro LC. Bladder paraganglioma: a case report. Pan Afr Med J 2020;36:339. [Crossref] [PubMed]
- Aksakal N, Agcaoglu O, Sahbaz NA, et al. Predictive Factors of Operative Hemodynamic Instability for Pheochromocytoma. Am Surg 2018;84:920-3. [Crossref] [PubMed]
- Seo SH, Kim JH, Kim MJ, et al. Whole Exome Sequencing Identifies Novel Genetic Alterations in Patients with Pheochromocytoma/Paraganglioma. Endocrinol Metab (Seoul) 2020;35:909-17. [Crossref] [PubMed]
- Lenders JW, Duh QY, Eisenhofer G, et al. Pheochromocytoma and paraganglioma: an endocrine society clinical practice guideline. J Clin Endocrinol Metab 2014;99:1915-42. [Crossref] [PubMed]
- Nölting S, Ullrich M, Pietzsch J, et al. Current Management of Pheochromocytoma/Paraganglioma: A Guide for the Practicing Clinician in the Era of Precision Medicine. Cancers (Basel) 2019;11:1505. [Crossref] [PubMed]



