
Prognostic value of LAMP1 in surgically resected esophageal squamous cell carcinoma
Introduction
Esophageal cancer (EC) is a common malignant tumor. And esophageal squamous cell carcinoma (ESCC) accounted for 90% in China (1,2). The 5-year survival rate of ESCC remains very low (3), which was negatively related to tumor invasion and metastasis (4). And less than 30% of ESCC patients can achieve early diagnosis and treatment.
Lysosomal associated membrane proteins (LAMPs), including LAMP1 and LAMP2, are major protein components of lysosome (5-7), which plays an important role in tumor evolution (8,9). Previous studies confirmed that LAMP1 showed high expression in astrocytoma (10). Furthermore, LAMP1 has also been found expressing on the cell surface of highly metastatic tumor cells, suggesting a role for LAMP1 in tumor cells migration (11-13).
In our previous study, we detected the LAMP1 expression in the surgically resected tissues of 584 patients with ESCC (14), which has been published on the “Pathology - Research and Practice” journal. The results showed that the LAMP1 expression levels were significantly different between TNM stages and degrees of histological differentiation, and that LAMP1 expression levels were negatively correlated with degrees of histological differentiation. But, one flaw is that our previous research is only limited to staging and histopathological classification without patient’s prognosis. In present study, we conducted a telephone follow-up of all the patients, and statistical analysis was carried out.
Methods
Patients’ information
Five hundred and eighty-four ESCC patients, included in our previous study (14), were followed up by the Follow-up Center of Affiliated Hospital of Jining Medical University. Patients’ clinical information and telephone number were collected from medical records. This study was reviewed and approved by the Medical Ethics Committee of Jining Medical University (2017-FY-007). Informed consent was obtained from all patients.
Immunohistochemistry (IHC)
Experimental procedures and criteria for determining results have been reported in our previous study. And the results of IHC were divided into four grades: “−”, “+”, “++”, and “+++”, which has been reported in our previous study (14). Except for the IHC results, all data in this study did not appear in our previous study. That is to say, we supplemented the patient’s prognostic information on the basis of existing immunohistochemical staining results (Figure 1).
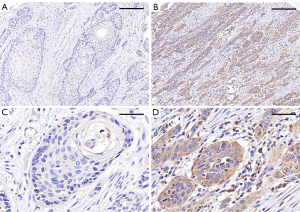
Statistical analysis
Logistic regression analysis and Kaplan-Meier survival curves were performed using SPSS software 22.0. P<0.05 was considered as statistically significant.
Results
Clinical data
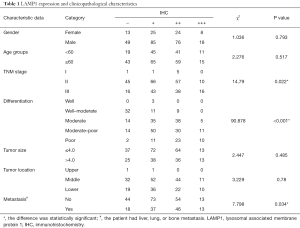
After telephone follow-up, we obtained prognostic information for only 298 ESCC patients. In addition, 7 cases of the 298 ESCC patients were only known to have died, but there was no accurate survival time. The remaining patients failed to get in touch. We calculated the patient’s survival rate simply according to the patient’s survival, on the basis of the deletion of a later collection of specimens. The 3-year survival rate was 54.34% (119/219), and the 5-year survival rate was 38.94% (44/113). The patients’ information and IHC results are listed in Table 1.
Full table
Data consistency
Before the new data analysis, we first compared the previous data with the existing data. In the existing data, the correlation analysis between the expression levels of LAMP1 and the degrees of differentiation was consistent with our previous paper. Similar results were also found in the analysis of TNM staging and other clinicopathological factors (data not shown).
Logistic regression analysis
Binary logistic regression analysis between LAMP1 expression and patients’ information (TNM stage, degrees of differentiation, gender, age, tumor location, metastasis, and tumor size) were performed. In this analysis, the positive degrees of LAMP1 were redefined. “−” and “+” were defined as the low expression. “++” and “+++” were defined as high expression (15). The results showed that the expression levels of LAMP1 were only correlated with degrees of differentiation. Further analysis showed that the degrees of differentiation showed medium negative correlation with LAMP1 expression with statistical significance (ORdifferentiation =0.502, P<0.001), while the correlation between another patients’ information (TNM stage, gender, age, tumor location, metastasis, and tumor size) and LAMP1 expression were not statistically significant (P>0.05).
Survival analysis
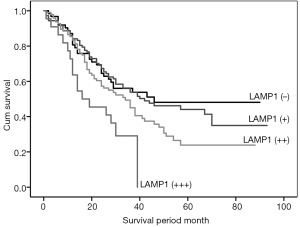
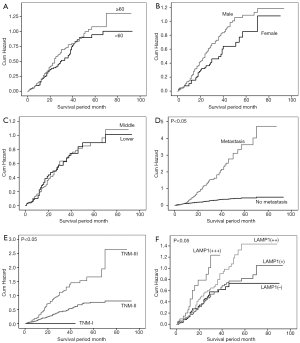
We made the Kaplan-Meier survival curves to analyze the correlations between LAMP1 expression levels and patients’ prognosis. Kaplan-Meier survival curves indicated that patients with a higher LAMP1 expression level showed worse prognosis (Figure 2, P<0.05). Additionally, we also analyzed the prognostic risk of LAMP1 expression levels and clinical features (age, gender, tumor location, organ metastasis, and TNM stage). The hazard plots showed that organ metastasis, high TNM classification, and high LAMP1 expression level were risk factors for survival in patients with ESCC (Figure 3, P<0.05), but age, gender, and tumor location not.
Discussion
LAMP1 is localized at the lysosomal membrane under physiological conditions (16,17). Previous studies showed that LAMP1 has been identified expressing higher in many cancers, especially in the metastatic cancer cells (11,13), which suggested that LAMP1 may be involved in the tumor invasion and metastasis.
In the present study, binary logistic regression analysis showed that LAMP1 expression was negatively correlated with degrees of tumor histological differentiation. The relationship between LAMP1 and tissue differentiation has been reported previously, which may be related to the high glycosylation of LAMP1 (18,19). Kaplan-Meier survival curves and hazard plots showed that high expression of LAMP1 is one of the risk factors for survival in patients with ESCC. The higher the expression of LAMP1, the worse the prognosis of ESCC patients. We will continue to study its roles and molecular mechanism in the following research.
The correlation between LAMP1 expression levels and TNM staging is inconsistent with our previous study. To test this inconsistency, we also made a Spearman Correlation Coefficient of the data for this study, which is consistent with our previous papers (data not shown). Furtherly, we also performed a logistics regression analysis of the IHC results and TNM stages in all 584 patients, which is also inconsistent with Spearman Correlation Coefficient analysis (data not shown). In short, logistics regression analysis is inconsistent with Spearman Correlation Coefficient analysis in this study. We think it may be caused by the following reasons: (I) sensitivity differences between the two analysis methods. Although Spearman correlation coefficient has statistical significance, but its value is relatively small; (II) the small number of some subgroups (Table 1). We planned to follow up 584 patients, but a large portion of the follow-up (nearly 50%) failed to be reached. This leads to the fact that our sample size is greatly reduced, and the accuracy of statistical analysis is affected to some extent. This limited sample number may influence the statistical analysis. On the other hand, many patients who were not contacted were likely to have died, which led to the discontinuation of the telephone number. We speculate that it could bias our final results. Perhaps there were more death patients in the population we couldn’t contact. So, our results still need to be further validated.
In conclusion, we have demonstrated that LAMP1 expression levels correlated with tumor histological differentiation and patients’ prognosis. High expression of LAMP1 predicts poor prognosis in ESCC patients.
Acknowledgments
Funding: This study was supported by the Medical Science and Technology Development Project of Shandong Province (2016WS0173) and Science and Technology Research Project of Shandong Higher Education Institutions (J13LK140), China.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. This study was conducted in accordance with the Declaration of Helsinki (as revised in 2013). This study was reviewed and approved by the Medical Ethics Committee of Jining Medical University (2017-FY-007). Informed consent was obtained from all patients.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Chen W, Zheng R, Baade PD, et al. Cancer statistics in China, 2015. CA Cancer J Clin 2016;66:115-32. [Crossref] [PubMed]
- Torre LA, Bray F, Siegel RL, et al. Global cancer statistics, 2012. CA Cancer J Clin 2015;65:87-108. [Crossref] [PubMed]
- Bao Y, Liu S, Zhou Q, et al. Three-dimensional conformal radiotherapy with concurrent chemotherapy for postoperative recurrence of esophageal squamous cell carcinoma: clinical efficacy and failure pattern. Radiat Oncol 2013;8:241. [Crossref] [PubMed]
- Rice TW, Rusch VW, Apperson-Hansen C, et al. Worldwide esophageal cancer collaboration. Dis Esophagus 2009;22:1-8. [Crossref] [PubMed]
- Saftig P, Klumperman J. Lysosome biogenesis and lysosomal membrane proteins: trafficking meets function. Nat Rev Mol Cell Biol 2009;10:623-35. [Crossref] [PubMed]
- Schroder BA, Wrocklage C, Hasilik A, et al. The proteome of lysosomes. Proteomics 2010;10:4053-76. [Crossref] [PubMed]
- Hook G, Jacobsen JS, Grabstein K, et al. Cathepsin B is a New Drug Target for Traumatic Brain Injury Therapeutics: Evidence for E64d as a Promising Lead Drug Candidate. Front Neurol 2015;6:178. [Crossref] [PubMed]
- Hamalisto S, Jaattela M. Lysosomes in cancer-living on the edge (of the cell). Curr Opin Cell Biol 2016;39:69-76. [Crossref] [PubMed]
- Liao X, Chen Y, Liu D, et al. High Expression of LAMP3 Is a Novel Biomarker of Poor Prognosis in Patients with Esophageal Squamous Cell Carcinoma. Int J Mol Sci 2015;16:17655-67. [Crossref] [PubMed]
- Jensen SS, Aaberg-Jessen C, Christensen KG, et al. Expression of the lysosomal-associated membrane protein-1 (LAMP-1) in astrocytomas. Int J Clin Exp Pathol 2013;6:1294-305. [PubMed]
- Agarwal AK, Srinivasan N, Godbole R, et al. Role of tumor cell surface lysosome-associated membrane protein-1 (LAMP1) and its associated carbohydrates in lung metastasis. J Cancer Res Clin Oncol 2015;141:1563-74. [Crossref] [PubMed]
- Saitoh O, Wang WC, Lotan R, et al. Differential glycosylation and cell surface expression of lysosomal membrane glycoproteins in sublines of a human colon cancer exhibiting distinct metastatic potentials. J Biol Chem 1992;267:5700-11. [PubMed]
- Agarwal AK, Gude RP, Kalraiya RD. Regulation of melanoma metastasis to lungs by cell surface Lysosome Associated Membrane Protein-1 (LAMP1) via galectin-3. Biochem Biophys Res Commun 2014;449:332-7. [Crossref] [PubMed]
- Huang J, Li L, Liu J, et al. Altered expression of lysosomal associated membrane protein 1 in esophageal squamous cell carcinoma. Pathol Res Pract 2017;213:938-42. [Crossref] [PubMed]
- Li L, Wang W, Zhang R, et al. High expression of LAMP2 predicts poor prognosis in patients with esophageal squamous cell carcinoma. Cancer Biomark 2017;19:305-11. [Crossref] [PubMed]
- Hasilik A, Wrocklage C, Schroder B. Intracellular trafficking of lysosomal proteins and lysosomes. Int J Clin Pharmacol Ther 2009;47:S18-33. [PubMed]
- Xu H, Ren D. Lysosomal physiology. Annu Rev Physiol 2015;77:57-80. [Crossref] [PubMed]
- Sarafian V, Jans R, Poumay Y. Expression of lysosome-associated membrane protein 1 (Lamp-1) and galectins in human keratinocytes is regulated by differentiation. Arch Dermatol Res 2006;298:73-81. [Crossref] [PubMed]
- Tyleckova J, Valekova I, Zizkova M, et al. Surface N-glycoproteome patterns reveal key proteins of neuronal differentiation. J Proteomics 2016;132:13-20. [Crossref] [PubMed]


