Prognostic value of tumor length in predicting survival for patients with esophageal cancer
Introduction
Esophageal cancer, which caused a total of 400,200 deaths in 2012, is the sixth leading cause of cancer death worldwide (1). Despite recent improvements in multimodal treatment, the 5-year relative survival rate was 41% for patients with localized stage, 23% for patients with regional stage, and only 5% for patients with distant stage esophageal cancer in the United States (2). Thus, an effective staging system is crucial for esophageal cancer patients in terms of determining the proper multidisciplinary therapy and estimating prognosis. Currently the T classification of the 7th TNM staging system (3) depends only on the depth of the primary tumor; furthermore, the 8th edition (4), which was placed into effect in January 2018, still does not consider tumor length. Several authors have recently investigated the importance of tumor length on overall survival (OS) for esophageal cancer patients (5-10). However, almost all of these studies focused on patients with early or regional stage cancer; patients with distant stage cancer were not included. In addition, most conclusions were analyzed based on a small number of patients.
Our study was conducted to explore the prognostic value of tumor length using a population-based study cohort; patient information was retrieved from the Surveillance, Epidemiology, and End Results (SEER) database. As previous studies reported that 3 cm was an adequate separation (5,7,11-14), we used this length to evaluate survival time.
Methods
Patients
The SEER Program of the National Cancer Institute provides information in a cancer-related database. Last released in April 2017, SEER-18 includes 18 cancer registries and covers 9,675,661 cancer cases from the US population diagnosed between 1973 and 2014 (15). Patients with a diagnosis of esophageal cancer were identified from the SEER database, using the SEER*Stat software (version 8.3.4). The reference number obtained to access research data files was 12631-Nov2016. Given the lack of personal identifying information, informed consent was waived. This study was approved by the ethics committee of The First Affiliated Hospital of Nanjing Medical University.
The inclusion criteria were as follows: (I) patients with Site recode ICD-O-3/WHO 2008 restricted to the “esophagus”; (II) exact tumor location (C15.0–C15.5); (III) diagnosed from 2010 or later. The exclusion criteria were as follows: unknown age, gender, race, tumor grade, tumor length, survival months, or TNM stage. Histology and tumor grade were coded based on the International Classification of Disease for Oncology, edition ICD-O-3. Tumor stage was coded according to the 7th edition of the TNM staging system.
Patients were categorized according to their ages (<65 and ≥65 years), gender (male and female), race (white, black and other), tumor grade [1–4], tumor location (cervical and upper third of esophagus; thoracic and middle third of the esophagus; abdominal and lower third of the esophagus), tumor length (≤3 and >3 cm), T stage [1–4], N stage [0–3] and M stage [0,1]. Histology was classified into three groups: esophageal adenocarcinoma (EAC, ICD-O-3 codes: 8140), esophageal squamous cell carcinoma (ESCC, ICD-O-3 codes: 8070) and others.
Statistical analysis
We divided the enrolled patients into two groups: a tumor length ≤3 cm group and a tumor length >3 cm group. Comparisons of the clinical characteristics between the two groups were performed using the chi-square test. OS of patients with different tumor lengths was evaluated using the Kaplan-Meier method and estimated by the log-rank test. Univariate and multivariate analyses were generated to examine the potential factors associated with OS. Hazard ratios (HRs) with 95% confidence intervals were used to quantify the strength of the association. Finally, a Cox proportional hazards analysis was performed to determine the association of tumor length with OS based on different T stages. A value of P<0.05 was defined as statistically significant. All analyses were performed using SPSS statistical software package (version 22.0).
Results
Patient characteristics
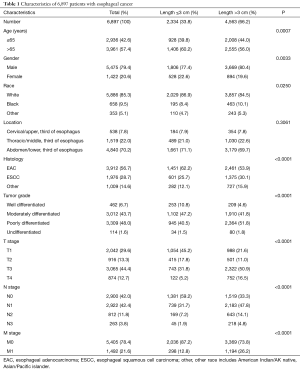
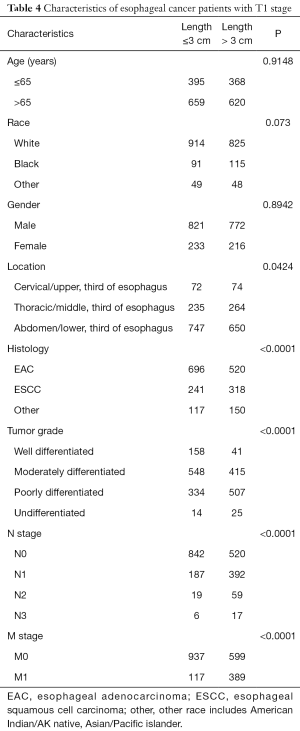
In total, 6,897 patients with esophageal cancer were identified, including 2,334 individuals with a tumor length ≤3 cm and 4,563 individuals with a tumor length >3 cm. The detailed clinical characteristics of the patients are shown in Table 1. The comparison between the two groups showed significant differences in all clinical characteristics (except tumor location). Patients with a tumor length ≤3 cm were more likely to have EAC (62.2% vs. 53.9%). Furthermore, with respect to tumor grade, patients with a tumor length ≤3 cm showed a better differentiation (10.8% vs. 4.6% in grade 1, 47.2% vs. 41.8% in grade 2). In addition, a significantly higher proportion of patients with a tumor length ≤3 cm was of earlier T stage (T1, T2), N stage (N0) and M stage (M0) (P<0.001).
Full table
Survival analysis
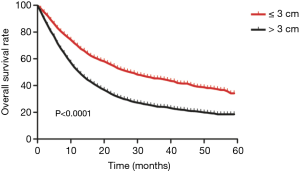
Kaplan-Meier analysis showed that patients with a tumor length ≤3 cm had a significantly better OS than those with a tumor length >3 cm (χ2=275.850, P=0.0001, Figure 1). The median survival time in the two groups was 27 months (95% CI: 24.1–29.9) and 12 months (95% CI: 11.4–12.6), respectively.
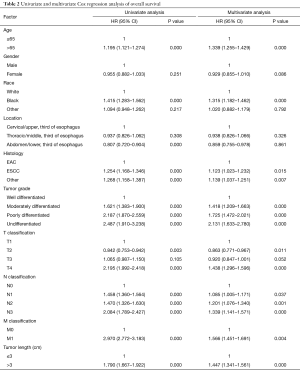
The details of the univariate and multivariate analysis on OS are shown in Table 2. Univariate analysis showed that patients with a tumor length >3 cm had a significantly greater risk of mortality compared than those with a tumor length ≤3 cm (HR: 1.790; 95% CI: 1.667–1.922; P=0.000). However, analysis of the T stage revealed that when T1 stage was taken as the reference, patients with T2 stage had better prognosis (HR: 0.842; 95% CI: 0.753–0.942; P=0.003), and no significant difference in OS was observed between T1 and T3 stage patients (HR: 1.065; 95% CI: 0.987–1.150; P=0.105). Furthermore, multivariate analysis demonstrated that patients with a tumor length >3 cm presented with poorer prognosis (HR: 1.447; 95% CI: 1.341-1.561; P=0.000) and T2 stage patients showed a lower risk of mortality (HR: 0.863; 95% CI: 0.771–0.967; P=0.011). Age, race, histology, tumor grade, N stage and M stage were also evaluated as independent prognostic factors for OS in the multivariate analysis (P<0.05).
Full table
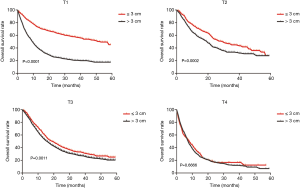
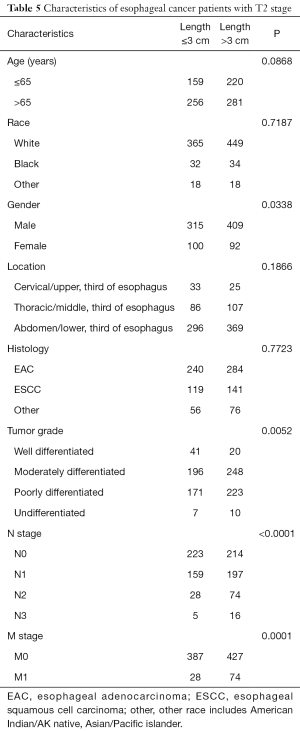
Finally, stratified analysis based on different T stages was performed to identify the prognostic importance of tumor length, and the details are shown in Table 3. Characteristics of esophageal cancer patients with T1 and T2 stage are shown in Tables 4 and 5 separately.

Full table
Full table
Full table
Univariate analysis indicated that compared with the tumor length ≤3 cm group, tumor length >3 cm group had poorer OS among T1 (HR: 2.869; 95% CI: 2.528–3.257; P=0.000), T2 (HR: 1.430; 95% CI: 1.180–1.732; P=0.000) and T3 (HR: 1.207; 95% CI: 1.079–1.350; P=0.001) stage patients but not among T4 stage patients (HR: 1.057; 95% CI: 0.851–1.312; P=0.617). When adjusted for age, gender, race, tumor location, tumor grade, histology, N stage and M stage, similar results were observed for T1 (HR: 1.844; 95% CI: 1.606–2.116; P=0.000), T2 (HR: 1.227; 95% CI: 1.003–1.501; P=0.046) and T3 (HR: 1.157; 95% CI: 1.033–1.295; P=0.012) stage patients by the multivariate Cox regression model. Survival curves of the tumor length groups based on different T stages are shown in Figure 2.
Discussion
Esophageal tumor length was used as a staging factor for the 1983 version of the AJCC TNM staging system (16). However, esophageal tumor length was replaced by depth of esophageal wall invasion in the 1987 version (17) because some studies demonstrated that the depth of tumor invasion more accurately correlated with survival than tumor length (18). From 1987 to the present, only the depth of tumor invasion has been used in the T classification. The current 7th edition of the UICC-AJCC TNM staging system includes the depth of tumor invasion, the number of positive lymph nodes, histology (adenocarcinoma and squamous cell carcinoma are staged separately), tumor grade and primary tumor location. However, tumor length is not included.
Some researchers have reported that tumor length is closely related to esophageal cancer outcome. Hollis et al retrospectively analyzed 389 esophageal cancer patients and demonstrated that tumor length on pathology was significantly related to OS, although no significant independent association was detected after adjusting for tumor-related factors (19). A similar finding was reported in a German study (20). However, other reports showed that tumor length may provide additional prognostic information beyond the TNM staging system, especially among esophageal cancer patients with early stages (7,18). Eloubeidi et al. conducted a population-based study, and they proposed tumor length as an independent prognostic factor in patients with localized disease; thus, they suggested a revised T-category of the esophageal TNM staging system to incorporate tumor length (9). Although this study provided some important evidence and was the largest study cohort, there were still some limitations. First, patients in their study were diagnosed in 1988 or later. However, clinical treatments and outcomes of esophageal cancer have recently improved, thereby compromising the strength of the evidence. Second, patients were staged using the 6th TNM edition, and thus, their results could not differentiate as to whether tumor length supplies additional prognostic information beyond the current 7th edition.
In the present study, we attempted to predict the outcomes of 6897 esophageal cancer patients diagnosed in 2010–2014 with known tumor length from the SEER database. All patients were staged according to the 7th edition. We found that patient age, race, histology, tumor grade, T stage, N stage, M stage and tumor length were all independent prognostic factors for OS. Patients with a tumor length ≤3 cm were more likely to be T1–2 (63%), N0 (59.2%) and M0 (87.2%) stage. The results of the multivariate Cox regression analysis also showed that tumor length >3 cm was associated with a worse OS among all 6,897 patients (HR: 1.447; 95% CI: 1.341–1.561; P=0.000).
The number of positive regional lymph nodes has been established as an independent predictor for esophageal cancers (21,22). Analysis of N classification in our study showed that mortality increased with progression of the N stage, consistent with the above reports. The depth of tumor invasion is another definite predictor for esophageal cancer. However, our results showed that T2 (HR: 0.863; 95% CI: 0.771–0.967; P=0.011) was associated with a lower risk of mortality compared with the T1 group, similar to the outcomes of Eloubeidi et al. (9). Interestingly, Wang et al. reported that there was no significant survival difference between T3 and T4 stages (21). These findings indicate that the current T stage, which is only based on depth of tumor invasion, may not be a good predictor for survival. In our stratified analysis, we classified all patients into T1, T2, T3 or T4 groups. The results reflected that the prognostic value of tumor length on OS was greatest among T1 stage patients (HR: 1.844; P=0.000) and weaker among T2 (HR: 1.227; P=0.046) and T3 (HR: 1.157; P=0.012) stage patients compared with T4 stage patients (HR: 1.057; P=0.617). Thus, the discriminatory power of tumor length was lost. We conclude that a tumor length >3 cm is closely related to an increased risk of mortality, especially when the tumor is localized within the lamina propria, the muscularis propria or the submucosa of the esophageal wall. When the primary tumor has developed over the esophageal wall, other factors determine the outcome.
Strengths of our study include the large number of patients from different institutions across the country and that all patients were staged according to the 7th edition of the AJCC-TNM staging system. However, there are still some limitations of our investigation. First, this is a retrospective study. Second, tumor length can be measured using various methods, including endoscopy (23,24), measurements by the pathologist, contrast esophagography, and CT scans. Thus, there may have been a lack of uniformity in measurement methods. Third, there were no details on treatment modalities, including techniques of surgical management, type of chemotherapy or radiotherapy. Each of these limitations may compromise the strength of the evidence.
In conclusion, our study suggests that esophageal tumor length is of great prognostic value in predicting OS, especially in early T stage patients. We propose that further prospective trials are needed to validate the prognostic value of tumor length among esophageal cancer patients.
Acknowledgments
Funding: This work was supported by funds from the National Natural Science Foundation of China (No. 81472809, 81502653, 81672983, 81703028), “333” Project of Jiangsu Province (No. BRA2012210), the Priority Academic Program Development of Jiangsu Higher Education Institutions (PAPD) (No. JX10231801), and the Six Major Talent Peak Project of Jiangsu Province (No. 2013-WSN-040).
Footnote
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/tcr.2018.05.07). The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. The study was conducted in accordance with the Declaration of Helsinki (as revised in 2013). Institutional ethical approval and informed consent were waived.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Torre LA, Bray F, Siegel RL, et al. Global cancer statistics, 2012. CA Cancer J Clin 2015;65:87-108. [Crossref] [PubMed]
- Siegel RL, Miller KD, Jemal A. Cancer statistics, 2017. CA Cancer J Clin 2017;67:7-30. [Crossref] [PubMed]
- Edge SB, Compton CC. The American Joint Committee on Cancer: the 7th Edition of the AJCC Cancer staging Manual and the Future of TNM. Ann Surg Oncol 2010;17:1471-4.
- Rice TW, Ishwaran H, Ferguson MK, et al. Cancer of the Esophagus and Esophagogastric Junction: An Eighth Edition Staging Primer.J Thorac Oncol 2017;12:36-42.
- Valmasoni M, Pierobon ES, Ruol A, et al. Endoscopic Tumor Length Should Be Reincluded in the Esophageal Cancer Staging System: Analyses of 662 Consecutive Patients. PLos One 2016;11:e0153068 [Crossref] [PubMed]
- Haisley KR, Hart KD, Fischer LE, et al. Increasing tumor length is associated with regional lymph node metastases and decreased survival in esophageal cancer. Am J Surg 2016;211:860-6. [Crossref] [PubMed]
- Song Z, Wang JW, Lin BC, et al. Analysis of the tumor length and other prognosis factors in pT1-2 node-negative esophageal squamous cell carcinoma in a Chinese population. World J Surg Oncol 2012;10:273. [Crossref] [PubMed]
- Gaur P, Sepesi B, Hofstetter WL, et al. Endoscopic esophageal tumor length: a prognostic factor for patients with esophageal cancer. Cancer 2011;117:63-9. [Crossref] [PubMed]
- Eloubeidi MA, Desmond R, Arguedas MR, et al. Prognostic factors for the survival of patients with esophageal carcinoma in the U.S.: the importance of tumor length and lymph node status. Cancer 2002;95:1434-43. [Crossref] [PubMed]
- Mirinezhad SK, Jangjoo AG, Seyenejad F, et al. Impact of Tumor Length on Survival for Patients with Resected Esophageal Cancer. Asian Pac J Cancer Prev 2014;15:691-4. [Crossref] [PubMed]
- Yendamuri S, Swisher SG, Correa AM, et al. Esophageal tumor length is independently associated with long-term survival. Cancer 2009;115:508-16. [Crossref] [PubMed]
- Zeybek A, Erdoğan A, Gülkesen KH, et al. Significance of tumor length as prognostic factor for esophageal cancer. Int Surg 2013;98:234-40. [Crossref] [PubMed]
- Berriochoa CA, Hibbard D, Morcos M, et al. Tumor length as a prognostic factor in esophageal cancer management. J Radiat Oncol 2015;4:71-7. [Crossref]
- Rizk N, Venkatraman E, Park B, et al. The prognostic importance of the number of involved lymph nodes in esophageal cancer: implications for revisions of the American Joint Committee on Cancer staging system. J Thorac Cardiovasc Surg 2006;132:1374-81. [Crossref] [PubMed]
- SEER: SEER*Stat Database: Incidence-SEER 18 Regs Research Data + Hurricane Katrina Impacted Louisiana Cases, Nov 2016 Sub (1973-2014 varying). National Cancer Institute, Surveillance, Epidemiology, and End Results Program. Available online: http://www.seer.cancer.gov
- Thompson WM. Esophageal cancer. Int J Radiat Oncol Biol Phys 1983;9:1533-65. [Crossref] [PubMed]
- Sobin LH, Hermanek P, Hutter RVP. TNM classification of malignant tumors: a comparison between the new (1987) and the old editions. Cancer 1988;61:2310-4. [Crossref] [PubMed]
- Wang BY, Goan YG, Hsu PK, et al. Tumor length as a prognostic factor in esophageal squamous cell carcinoma. Ann Thorac Surg 2011;91:887-93. [Crossref] [PubMed]
- Hollis AC, Quinn LM, Hodson J, et al. Prognostic significance of tumor length in patients receiving esophagectomy for esophageal cancer. J Surg Oncol 2017;116:1114-22. [Crossref] [PubMed]
- Bollschweiler E, Baldus SE, Schröder W, et al. Staging of esophageal carcinoma: length of tumor and number of involved regional lymph nodes. Are these independent prognostic factors? J Surg Oncol 2006;94:355-63. [Crossref] [PubMed]
- Wang J, Wu N, Zheng QF, et al. Evaluation of the 7th edition of the TNM classification in patients with resected esophageal squamous cell carcinoma. World J Gastroenterol 2014;20:18397-403.
- Twine CP, Roberts SA, Lewis WG, et al. Prognostic significance of endoluminal ultrasound-defined disease length and tumor volume (EDTV) for patients with the diagnosis of esophageal cancer. Surg Endosc 2010;24:870-8. [Crossref] [PubMed]
- Bhutani MS, Barde CJ, Markert RJ, et al. Length of Esophageal Cancer and Degree of Luminal Stenosis during Upper Endoscopy Predict T stage by Endoscopic Ultrasound. Endoscopy 2002;34:461-63. [Crossref] [PubMed]
- Bolton WD, Hofstetter WL, Francis AM, et al. Impact of tumor length on long-term survival of pT1 esophageal adenocarcinoma. J Thorac Cardiovasc Surg 2009;138:831-6. [Crossref] [PubMed]